Naked2 acts as a cargo recognition and targeting protein to ensure proper delivery and fusion of TGF-alpha containing exocytic vesicles at the lower lateral membrane of polarized MDCK cells
- PMID: 17553928
- PMCID: PMC1949375
- DOI: 10.1091/mbc.e07-02-0172
Naked2 acts as a cargo recognition and targeting protein to ensure proper delivery and fusion of TGF-alpha containing exocytic vesicles at the lower lateral membrane of polarized MDCK cells
Abstract
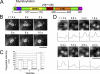
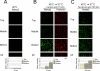
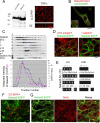
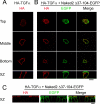
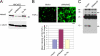
Transforming growth factor-alpha (TGF-alpha) is the major autocrine EGF receptor ligand in vivo. In polarized epithelial cells, proTGF-alpha is synthesized and then delivered to the basolateral cell surface. We previously reported that Naked2 interacts with basolateral sorting determinants in the cytoplasmic tail of a Golgi-processed form of TGF-alpha and that TGF-alpha is not detected at the basolateral surface of Madin-Darby canine kidney (MDCK) cells expressing myristoylation-deficient (G2A) Naked2. By high-resolution microscopy, we now show that wild-type, but not G2A, Naked2-associated vesicles fuse at the plasma membrane. We further demonstrate that Naked2-associated vesicles are delivered to the lower lateral membrane of polarized MDCK cells independent of mu1B adaptin. We identify a basolateral targeting segment within Naked2; residues 1-173 redirect NHERF-1 from the apical cytoplasm to the basolateral membrane, and internal deletion of residues 37-104 results in apical mislocalization of Naked2 and TGF-alpha. Short hairpin RNA knockdown of Naked2 leads to a dramatic reduction in the 16-kDa cell surface isoform of TGF-alpha and increased cytosolic TGF-alpha immunoreactivity. We propose that Naked2 acts as a cargo recognition and targeting (CaRT) protein to ensure proper delivery, tethering, and fusion of TGF-alpha-containing vesicles to a distinct region at the basolateral surface of polarized epithelial cells.
Figures
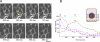

Similar articles
-
Use of fluorescence-activated vesicle sorting for isolation of Naked2-associated, basolaterally targeted exocytic vesicles for proteomics analysis.Mol Cell Proteomics. 2008 Sep;7(9):1651-67. doi: 10.1074/mcp.M700155-MCP200. Epub 2008 May 25. Mol Cell Proteomics. 2008. PMID: 18504258 Free PMC article.
-
Myristoylated Naked2 escorts transforming growth factor alpha to the basolateral plasma membrane of polarized epithelial cells.Proc Natl Acad Sci U S A. 2004 Apr 13;101(15):5571-6. doi: 10.1073/pnas.0401294101. Epub 2004 Apr 2. Proc Natl Acad Sci U S A. 2004. PMID: 15064403 Free PMC article.
-
EGF receptor-independent action of TGF-alpha protects Naked2 from AO7-mediated ubiquitylation and proteasomal degradation.Proc Natl Acad Sci U S A. 2008 Sep 9;105(36):13433-8. doi: 10.1073/pnas.0806298105. Epub 2008 Aug 29. Proc Natl Acad Sci U S A. 2008. PMID: 18757723 Free PMC article.
-
Protein trafficking in the exocytic pathway of polarized epithelial cells.Trends Cell Biol. 2001 Dec;11(12):483-6. doi: 10.1016/s0962-8924(01)02145-6. Trends Cell Biol. 2001. PMID: 11719053 Review.
-
Membrane traffic in polarized epithelial cells.Curr Opin Cell Biol. 2000 Aug;12(4):483-90. doi: 10.1016/s0955-0674(00)00120-4. Curr Opin Cell Biol. 2000. PMID: 10873817 Review.
Cited by
-
High temperature requirement A3 (HtrA3) promotes etoposide- and cisplatin-induced cytotoxicity in lung cancer cell lines.J Biol Chem. 2010 Apr 16;285(16):12011-27. doi: 10.1074/jbc.M109.097790. Epub 2010 Feb 12. J Biol Chem. 2010. PMID: 20154083 Free PMC article.
-
Taking the scenic route: biosynthetic traffic to the plasma membrane in polarized epithelial cells.Traffic. 2009 Aug;10(8):972-81. doi: 10.1111/j.1600-0854.2009.00927.x. Epub 2009 May 12. Traffic. 2009. PMID: 19453969 Free PMC article. Review.
-
A monoclonal antibody produced against Naked2.Monoclon Antib Immunodiagn Immunother. 2013 Aug;32(4):290-4. doi: 10.1089/mab.2012.0111. Monoclon Antib Immunodiagn Immunother. 2013. PMID: 23909424 Free PMC article.
-
Clathrin and AP1B: key roles in basolateral trafficking through trans-endosomal routes.FEBS Lett. 2009 Dec 3;583(23):3784-95. doi: 10.1016/j.febslet.2009.10.050. Epub 2009 Oct 23. FEBS Lett. 2009. PMID: 19854182 Free PMC article. Review.
-
Trafficking of epidermal growth factor receptor ligands in polarized epithelial cells.Annu Rev Physiol. 2014;76:275-300. doi: 10.1146/annurev-physiol-021113-170406. Epub 2013 Nov 8. Annu Rev Physiol. 2014. PMID: 24215440 Free PMC article. Review.
References
-
- Bonifacino J. S., Glick B. S. The mechanisms of vesicle budding and fusion. Cell. 2004;116:153–166. - PubMed
-
- Bonifacino J. S., Traub L. M. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 2003;72:395–447. - PubMed
-
- Dempsey P. J., Coffey R. J. Basolateral targeting and efficient consumption of transforming growth factor-alpha when expressed in Madin-Darby canine kidney cells. J. Biol. Chem. 1994;269:16878–16889. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

