Localization and expression of group I metabotropic glutamate receptors in the mouse striatum, globus pallidus, and subthalamic nucleus: regulatory effects of MPTP treatment and constitutive Homer deletion
- PMID: 17553998
- PMCID: PMC6672159
- DOI: 10.1523/JNEUROSCI.3819-06.2007
Localization and expression of group I metabotropic glutamate receptors in the mouse striatum, globus pallidus, and subthalamic nucleus: regulatory effects of MPTP treatment and constitutive Homer deletion
Abstract
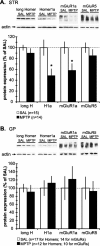
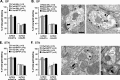
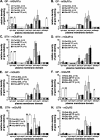
Group I metabotropic glutamate receptors (mGluRs), mGluR1 and mGluR5, regulate activity in the globus pallidus (GP) and subthalamic nucleus (STN). To test whether the localization of group I mGluRs is altered in parkinsonism, we used immunoelectron microscopy to analyze the subcellular and subsynaptic distribution of mGluR1a and mGluR5 in GP and STN of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated mice. Homer1 and Homer2 knock-out mice were used to assess the role of Homer in MPTP-induced redistribution of group I mGluRs. We also examined the effects of MPTP on the expression levels of group I mGluRs and Homer proteins in GP and striatum. MPTP treatment significantly reduced the expression levels of H1a and mGluR1a in striatum but not in GP. Although light microscopy did not reveal noticeable effects of MPTP treatment on the distribution of group I mGluRs and Homer proteins in GP and STN, specific changes in the ultrastructural localization of mGluR1a were found in MPTP-treated normal and Homer knock-out mice. An increase in the expression of presynaptic axonal and terminal mGluR1a labeling and an increased level of mGluR1a immunoreactivity in the postsynaptic specialization of putative GABAergic synapses were among the most significant effects induced by dopamine depletion. However, neither of these changes was found for mGluR5, which, in contrast, displayed complex regulatory alterations in its subsynaptic distribution in response to Homer deletion and MPTP lesion. Thus, nigrostriatal dopaminergic lesion and Homer deletion lead to changes in the trafficking of group I mGluRs in vivo that are specific to receptor subtypes and brain areas.
Figures




Similar articles
-
Changes in glutamate receptors in dyskinetic parkinsonian monkeys after unilateral subthalamotomy.J Neurosurg. 2015 Dec;123(6):1383-93. doi: 10.3171/2014.10.JNS141570. Epub 2015 May 1. J Neurosurg. 2015. PMID: 25932606
-
Group I metabotropic glutamate receptors at GABAergic synapses in monkeys.J Neurosci. 1999 Aug 1;19(15):6488-96. doi: 10.1523/JNEUROSCI.19-15-06488.1999. J Neurosci. 1999. PMID: 10414977 Free PMC article.
-
Subcellular and subsynaptic localization of group I metabotropic glutamate receptors in the monkey subthalamic nucleus.J Comp Neurol. 2004 Jul 5;474(4):589-602. doi: 10.1002/cne.20158. J Comp Neurol. 2004. PMID: 15174075
-
GABA(B) and group I metabotropic glutamate receptors in the striatopallidal complex in primates.J Anat. 2000 May;196 ( Pt 4)(Pt 4):555-76. doi: 10.1046/j.1469-7580.2000.19640555.x. J Anat. 2000. PMID: 10923987 Free PMC article. Review.
-
Group I metabotropic glutamate receptor-mediated gene expression in striatal neurons.Neurochem Res. 2008 Oct;33(10):1920-4. doi: 10.1007/s11064-008-9654-4. Epub 2008 Mar 20. Neurochem Res. 2008. PMID: 18351459 Review.
Cited by
-
Regulation of group I metabotropic glutamate receptor expression in the rat striatum and prefrontal cortex in response to amphetamine in vivo.Brain Res. 2010 Apr 22;1326:184-92. doi: 10.1016/j.brainres.2010.02.062. Epub 2010 Feb 26. Brain Res. 2010. PMID: 20193665 Free PMC article.
-
Subcellular and subsynaptic localization of group I metabotropic glutamate receptors in the nucleus accumbens of cocaine-treated rats.Neuroscience. 2008 Jun 23;154(2):653-66. doi: 10.1016/j.neuroscience.2008.03.049. Epub 2008 Mar 29. Neuroscience. 2008. PMID: 18479833 Free PMC article.
-
Imbalances in prefrontal cortex CC-Homer1 versus CC-Homer2 expression promote cocaine preference.J Neurosci. 2013 May 8;33(19):8101-13. doi: 10.1523/JNEUROSCI.1727-12.2013. J Neurosci. 2013. PMID: 23658151 Free PMC article.
-
Group I mGluRs evoke K-ATP current by intracellular Ca2+ mobilization in rat subthalamus neurons.J Pharmacol Exp Ther. 2013 Apr;345(1):139-50. doi: 10.1124/jpet.112.201566. Epub 2013 Jan 18. J Pharmacol Exp Ther. 2013. PMID: 23335392 Free PMC article.
-
Anatomical localization of Cav3.1 calcium channels and electrophysiological effects of T-type calcium channel blockade in the motor thalamus of MPTP-treated monkeys.J Neurophysiol. 2016 Jan 1;115(1):470-85. doi: 10.1152/jn.00858.2015. Epub 2015 Nov 4. J Neurophysiol. 2016. PMID: 26538609 Free PMC article.
References
-
- Awad-Granko H, Conn PJ. Activation of groups I or III metabotropic glutamate receptors inhibits excitatory transmission in the rat subthalamic nucleus. Neuropharmacology. 2001;41:32–41. - PubMed
-
- Betarbet R, Porter RH, Greenamyre JT. GluR1 glutamate receptor subunit is regulated differentially in the primate basal ganglia following nigrostriatal dopamine denervation. J Neurochem. 2000;74:1166–1174. - PubMed
-
- Bezard E, Dovero S, Bioulac B, Gross C. Effects of different schedules of MPTP administration on dopaminergic neurodegeneration in mice. Exp Neurol. 1997;148:288–292. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
