GAP-43 gene expression regulates information storage
- PMID: 17554085
- PMCID: PMC1896091
- DOI: 10.1101/lm.581907
GAP-43 gene expression regulates information storage
Abstract
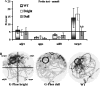
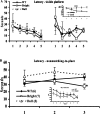
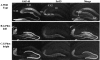
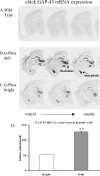
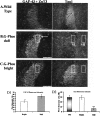
Previous reports have shown that overexpression of the growth- and plasticity-associated protein GAP-43 improves memory. However, the relation between the levels of this protein to memory enhancement remains unknown. Here, we studied this issue in transgenic mice (G-Phos) overexpressing native, chick GAP-43. These G-Phos mice could be divided at the behavioral level into "spatial bright" and "spatial dull" groups based on their performance on two hidden platform water maze tasks. G-Phos dull mice showed both acquisition and retention deficits on the fixed hidden platform task, but were able to learn a visible platform task. G-Phos bright mice showed memory enhancement relative to wild type on the more difficult movable hidden platform spatial memory task. In the hippocampus, the G-Phos dull group showed a 50% greater transgenic GAP-43 protein level and a twofold elevated transgenic GAP-43 mRNA level than that measured in the G-Phos bright group. Unexpectedly, the dull group also showed an 80% reduction in hippocampal Tau1 staining. The high levels of GAP-43 seen here leading to memory impairment find its histochemical and behavioral parallel in the observation of Rekart et al. (Neuroscience 126: 579-584) who described elevated levels of GAP-43 protein in the hippocampus of Alzheimer's patients. The present data suggest that moderate overexpression of a phosphorylatable plasticity-related protein can enhance memory, while excessive overexpression may produce a "neuroplasticity burden" leading to degenerative and hypertrophic events culminating in memory dysfunction.
Figures


Similar articles
-
Ectopic growth of hippocampal mossy fibers in a mutated GAP-43 transgenic mouse with impaired spatial memory retention.Hippocampus. 2010 Jan;20(1):58-64. doi: 10.1002/hipo.20635. Hippocampus. 2010. PMID: 19437419 Free PMC article.
-
In vivo post-transcriptional regulation of GAP-43 mRNA by overexpression of the RNA-binding protein HuD.J Neurochem. 2006 Feb;96(3):790-801. doi: 10.1111/j.1471-4159.2005.03607.x. Epub 2006 Jan 9. J Neurochem. 2006. PMID: 16405504
-
Hippocampal-dependent memory is impaired in heterozygous GAP-43 knockout mice.Hippocampus. 2005;15(1):1-7. doi: 10.1002/hipo.20045. Hippocampus. 2005. PMID: 15390153
-
Effects of age and spatial learning on adenylyl cyclase mRNA expression in the mouse hippocampus.Neurobiol Aging. 2004 Sep;25(8):1095-106. doi: 10.1016/j.neurobiolaging.2003.10.014. Neurobiol Aging. 2004. PMID: 15212834
-
Associative and spatial learning and memory deficits in transgenic mice overexpressing the RNA-binding protein HuD.Neurobiol Learn Mem. 2007 May;87(4):635-43. doi: 10.1016/j.nlm.2006.11.004. Epub 2006 Dec 20. Neurobiol Learn Mem. 2007. PMID: 17185008
Cited by
-
Effects of Prenatal Dexamethasone Treatment and Post-Weaning Moderate Fructose Intake on Synaptic Plasticity and Behavior in Adult Male Wistar Rat Offspring.Biology (Basel). 2024 Jul 19;13(7):547. doi: 10.3390/biology13070547. Biology (Basel). 2024. PMID: 39056739 Free PMC article.
-
Developmental and adult GAP-43 deficiency in mice dynamically alters hippocampal neurogenesis and mossy fiber volume.Dev Neurosci. 2014;36(1):44-63. doi: 10.1159/000357840. Epub 2014 Feb 26. Dev Neurosci. 2014. PMID: 24576816 Free PMC article.
-
Dopamine-mediated MK-801-induced elevation in food-based extinction responding in rats and associated changes in region-specific phosphorylated ERK.Psychopharmacology (Berl). 2010 Oct;212(3):393-403. doi: 10.1007/s00213-010-1959-8. Epub 2010 Jul 23. Psychopharmacology (Berl). 2010. PMID: 20652538
-
Hippocampal expression of synaptic structural proteins and phosphorylated cAMP response element-binding protein in a rat model of vascular dementia induced by chronic cerebral hypoperfusion.Neural Regen Res. 2012 Apr 15;7(11):821-6. doi: 10.3969/j.issn.1673-5374.2012.11.004. Neural Regen Res. 2012. PMID: 25737708 Free PMC article.
-
Cholinergic dysfunctions and enhanced oxidative stress in the neurobehavioral toxicity of lambda-cyhalothrin in developing rats.Neurotox Res. 2012 Nov;22(4):292-309. doi: 10.1007/s12640-012-9313-z. Epub 2012 Feb 11. Neurotox Res. 2012. PMID: 22327935
References
-
- Aigner L., Arber S., Kapfhammer J.P., Laux T., Schneider C., Botteri F., Brenner H.R., Caroni P. Overexpression of the neural growth-associated protein GAP-43 induces nerve sprouting in the adult nervous system of transgenic mice. Cell. 1995;83:269–278. - PubMed
-
- Alexander K.A., Wakim B.T., Doyle G.S., Walsh K.A., Storm D.R. Identification and characterization of the calmodulin-binding domain of neuromodulin, a neurospecific calmodulin-binding protein. J. Biol. Chem. 1988;263:7544–7549. - PubMed
-
- Baizer L., Alkan S., Stocker K., Ciment G. Chicken growth-associated protein (GAP)-43: Primary structure and regulated expression of mRNA during embryogenesis. Brain Res. Mol. Brain Res. 1990;7:61–68. - PubMed
-
- Basi G.S., Jacobson R.D., Virag I., Schilling J., Skene J.H. Primary structure and transcriptional regulation of GAP-43, a protein associated with nerve growth. Cell. 1987;49:785–791. - PubMed
-
- Benowitz L.I., Routtenberg A. A membrane phosphoprotein associated with neural development, axonal regeneration, phospholipid metabolism, and synaptic plasticity. Trends Neurosci. 1987;10:527–532.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous
