Correlation of quantitative MRI and neuropathology in epilepsy surgical resection specimens--T2 correlates with neuronal tissue in gray matter
- PMID: 17555988
- PMCID: PMC1971136
- DOI: 10.1016/j.neuroimage.2007.04.051
Correlation of quantitative MRI and neuropathology in epilepsy surgical resection specimens--T2 correlates with neuronal tissue in gray matter
Abstract
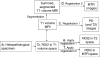
Newer MRI methods can detect cerebral abnormalities not identified on routine imaging in patients with focal epilepsy. Correlation of MRI with histopathology is necessary to understand the basis of MRI abnormalities and subsequently predict histopathology from in vivo MRI. The aim of this study was to determine if particular quantitative MR parameters were associated with particular histological features. Nine patients with temporal lobe epilepsy were imaged at 1.5 T using standard presurgical volumetric and quantifiable sequences: magnetization transfer and FFT2. The resected temporal lobe was registered with the volumetric MRI data according to our previously described method to permit correlation of the modalities. Stereologically measured neuronal densities and field fraction of GFAP, MAP2, synaptophysin and NeuN immunohistochemistry were obtained. Analyses were performed in the middle temporal gyrus and compared with quantitative MRI data from the equivalent regions. There was a significant Spearman Rho negative correlation between NeuN field fraction and the T2 value in gray matter (correlation coefficient -0.72, p=0.028). There were no significant correlations between any neuropathological and MR measures in white matter. These preliminary findings suggest that T2 in gray matter is sensitive to the proportion of neuronal tissue. Novel quantitative MRI measures acquired with higher field strength magnets, and so with superior signal to noise ratios, may generate data that correlate with histopathological measures. This will enable better identification and delineation of the structural causes of refractory focal epilepsy, and will be of particular benefit in patients in whom current optimal MRI does not identify a relevant abnormality.
Figures
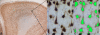



References
-
- Blume W.T., Ganapathy G.R., Munoz D., Lee D.H. Indices of resective surgery effectiveness for intractable nonlesional focal epilepsy. Epilepsia. 2004;45:46–53. - PubMed
-
- Brochet B., Dousset V. Pathological correlates of magnetization transfer imaging abnormalities in animal models and humans with multiple sclerosis. Neurology. 1999;53:S12–S17. - PubMed
-
- Cascino G.D., Jack C.R., Jr., Parisi J.E., Sharbrough F.W., Hirschorn K.A., Meyer F.B., Marsh W.R., O'Brien P.C. Magnetic resonance imaging-based volume studies in temporal lobe epilepsy: pathological correlations. Ann. Neurol. 1991;30:31–36. - PubMed
-
- Eriksson S.H., Free S.L., Thom M., Harkness W., Sisodiya S.M., Duncan J.S. Reliable registration of preoperative MRI with histopathology after temporal lobe resections. Epilepsia. 2005;46:1646–1653. - PubMed
-
- Eriksson S.H., Free S.L., Thom M., Martinian L., Sisodiya S.M. Methodological aspects of 3D and automated 2D analyses of white matter neuronal density in temporal lobe epilepsy. Neuropathol. Appl. Neurobiol. 2006;3:260–270. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

