Coordinated regulation of Toll-like receptor and NOD2 signaling by K63-linked polyubiquitin chains
- PMID: 17562858
- PMCID: PMC1952158
- DOI: 10.1128/MCB.00270-07
Coordinated regulation of Toll-like receptor and NOD2 signaling by K63-linked polyubiquitin chains
Abstract
K63 polyubiquitin chains spatially and temporally link innate immune signaling effectors such that cytokine release can be coordinated. Crohn's disease is a prototypical inflammatory disorder in which this process may be faulty as the major Crohn's disease-associated protein, NOD2 (nucleotide oligomerization domain 2), regulates the formation of K63-linked polyubiquitin chains on the I kappa kinase (IKK) scaffolding protein, NEMO (NF-kappaB essential modifier). In this work, we study these K63-linked ubiquitin networks to begin to understand the biochemical basis for the signaling cross talk between extracellular pathogen Toll-like receptors (TLRs) and intracellular pathogen NOD receptors. This work shows that TLR signaling requires the same ubiquitination event on NEMO to properly signal through NF-kappaB. This ubiquitination is partially accomplished through the E3 ubiquitin ligase TRAF6. TRAF6 is activated by NOD2, and this activation is lost with a major Crohn's disease-associated NOD2 allele, L1007insC. We further show that TRAF6 and NOD2/RIP2 share the same biochemical machinery (transforming growth factor beta-activated kinase 1 [TAK1]/TAB/Ubc13) to activate NF-kappaB, allowing TLR signaling and NOD2 signaling to synergistically augment cytokine release. These findings suggest a biochemical mechanism for the faulty cytokine balance seen in Crohn's disease.
Figures




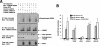




References
-
- Abbott, D. W., A. Wilkins, J. M. Asara, and L. C. Cantley. 2004. The Crohn's disease protein, NOD2, requires RIP2 in order to induce ubiquitinylation of a novel site on NEMO. Curr. Biol. 14:2217-2227. - PubMed
-
- Boone, D. L., E. E. Turer, E. G. Lee, R. C. Ahmad, M. T Wheeler, et al. 2004. The ubiquitin-modifying enzyme A20 is required for termination of Toll-like receptor responses. Nat. Immunol. 5:1052-1060. - PubMed
-
- Chung, J. Y., Y. C. Park, H. Ye, and H. Wu. 2002. All TRAFs are not created equal: common and distinct molecular mechanisms of TRAF-mediated signal transduction. J. Cell Sci. 115:679-688. - PubMed
-
- Deng, L., C. Wang, E. Spencer, L. Yang, A. Braun, et al. 2000. Activation of the IkappaB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell 103:351-361. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
