Nanosecond-timescale conformational dynamics of the human alpha7 nicotinic acetylcholine receptor
- PMID: 17573436
- PMCID: PMC1989720
- DOI: 10.1529/biophysj.107.109843
Nanosecond-timescale conformational dynamics of the human alpha7 nicotinic acetylcholine receptor
Abstract
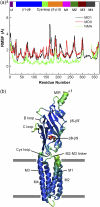
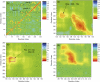
We explore the conformational dynamics of a homology model of the human alpha7 nicotinic acetylcholine receptor using molecular dynamics simulation and analyses of root mean-square fluctuations, block partitioning of segmental motion, and principal component analysis. The results reveal flexible regions and concerted global motions of the subunits encompassing extracellular and transmembrane domains of the subunits. The most relevant motions comprise a bending, hinged at the beta10-M1 region, accompanied by concerted tilting of the M2 helices that widens the intracellular end of the channel. Despite the nanosecond timescale, the observations suggest that tilting of the M2 helices may initiate opening of the pore. The results also reveal direct coupling between a twisting motion of the extracellular domain and dynamic changes of M2. Covariance analysis of interresidue motions shows that this coupling arises through a network of residues within the Cys and M2-M3 loops where Phe135 is stabilized within a hydrophobic pocket formed by Leu270 and Ile271. The resulting concerted motion causes a downward shift of the M2 helices that disrupts a hydrophobic girdle formed by 9' and 13' residues.
Figures

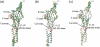






References
-
- Sine, S. M., and A. G. Engel. 2006. Recent advances in Cys-loop receptor structure and function. Nature. 440:448–455. - PubMed
-
- Celie, P. H., S. E. van Rossum-Fikkert, W. J. van Dijk, K. Brejc, A. B. Smit, and T. K. Sixma. 2004. Nicotine and carbamylcholine binding to nicotinic acetylcholine receptors as studied in AChBP crystal structures. Neuron. 41:907–914. - PubMed
-
- Gao, F., N. Bren, T. P. Burghardt, S. Hansen, R. H. Henchman, P. Taylor, J. A. McCammon, and S. M. Sine. 2005. Agonist-mediated conformational changes in acetylcholine-binding protein revealed by simulation and intrinsic tryptophan fluorescence. J. Biol. Chem. 280:8443–8451. - PubMed
-
- Shi, J., J. R. Koeppe, E. A. Komives, and P. Taylor. 2006. Ligand-induced conformational changes in the acetylcholine-binding protein analyzed by hydrogen-deuterium exchange mass spectrometry. J. Biol. Chem. 281:12170–12177. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

