Cholesterol depletion alters detergent-specific solubility profiles of selected tight junction proteins and the phosphorylation of occludin
- PMID: 17574235
- PMCID: PMC2169288
- DOI: 10.1016/j.yexcr.2007.05.009
Cholesterol depletion alters detergent-specific solubility profiles of selected tight junction proteins and the phosphorylation of occludin
Abstract
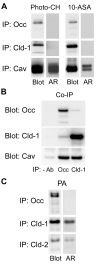
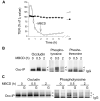
Differential centrifugation of Triton X-100 or CHAPS lysates from control and cholesterol (CH)-depleted MDCK II cells, segregated integral tight junction (TJ) proteins associated with detergent-resistant membranes (DRMs) into two groups. Group A proteins (occludin, claudin-2 and -3) were detected in large, intermediate and small aggregates in both detergents, whereas group B proteins (claudin-1, -4 and -7) were observed in small aggregates in TX-100 and in intermediate and small aggregates in CHAPS. Depletion of CH altered the distribution of group A and B proteins among the three size categories in a detergent-specific manner. In lysates produced with octyl glucoside, a detergent that selectively extracts proteins from DRMs, group A proteins were undetectable in large aggregates and CH depletion did not alter the distribution of either group A or B proteins in intermediate or small aggregates. Neither occludin (group A) nor claudin-1 (group B) was in intimate enough contact with CH to be cross-linked to [(3)H]-photo-cholesterol. However, antibodies to either TJ protein co-immunoprecipitated caveolin-1, a CH-binding protein. Unlike claudins, occludin's presence in TJs and DRMs did not require palmitoylation. Equilibrium density centrifugation on discontinuous OptiPrep gradients revealed detergent-related differences in the densities of TJ-bearing DRMs. There was little or no change in those densities after CH depletion. Removing CH from the plasma membrane increased tyrosine and threonine phosphorylation of occludin, and transepithelial electrical resistance (TER) within 30 min. After 2 h of CH efflux, phospho-occludin levels and TER fell below control values. We conclude that the association of integral TJ proteins with DRMS, pelleted at low speeds, is partially CH-dependent. However, the buoyant density of TJ-associated DRMs is a function of the detergent used and is insensitive to decreases in CH.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

