Dynamic assembly of chromatin complexes during cellular senescence: implications for the growth arrest of human melanocytic nevi
- PMID: 17578512
- PMCID: PMC1974778
- DOI: 10.1111/j.1474-9726.2007.00308.x
Dynamic assembly of chromatin complexes during cellular senescence: implications for the growth arrest of human melanocytic nevi
Abstract
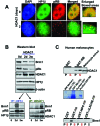
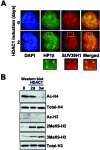
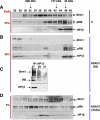
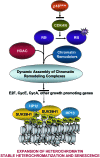
The retinoblastoma (RB)/p16(INK4a) pathway regulates senescence of human melanocytes in culture and oncogene-induced senescence of melanocytic nevi in vivo. This senescence response is likely due to chromatin modifications because RB complexes from senescent melanocytes contain increased levels of histone deacetylase (HDAC) activity and tethered HDAC1. Here we show that HDAC1 is prominently detected in p16(INK4a)-positive, senescent intradermal melanocytic nevi but not in proliferating, recurrent nevus cells that localize to the epidermal/dermal junction. To assess the role of HDAC1 in the senescence of melanocytes and nevi, we used tetracycline-based inducible expression systems in cultured melanocytic cells. We found that HDAC1 drives a sequential and cooperative activity of chromatin remodeling effectors, including transient recruitment of Brahma (Brm1) into RB/HDAC1 mega-complexes, formation of heterochromatin protein 1 beta (HP1 beta)/SUV39H1 foci, methylation of H3-K9, stable association of RB with chromatin and significant global heterochromatinization. These chromatin changes coincide with expression of typical markers of senescence, including the senescent-associated beta-galactosidase marker. Notably, formation of RB/HP1 beta foci and early tethering of RB to chromatin depends on intact Brm1 ATPase activity. As cells reached senescence, ejection of Brm1 from chromatin coincided with its dissociation from HP1 beta/RB and relocalization to protein complexes of lower molecular weight. These results provide new insights into the role of the RB pathway in regulating cellular senescence and implicate HDAC1 as a likely mediator of early chromatin remodeling events.
Figures



Comment in
-
Remodeling chromatin for senescence.Aging Cell. 2007 Aug;6(4):425-7. doi: 10.1111/j.1474-9726.2007.00313.x. Aging Cell. 2007. PMID: 17635419
References
-
- Aagaard L, Laible G, Selenko P, Schmid M, Dorn R, Schotta G, Kuhfittig S, Wolf A, Lebersorger A, Singh PB, Reuter G, Jenuwein T. Functional mammalian homologues of the Drosophila PEV-modifier Su(var)3-9 encode centromere-associated proteins which complex with the heterochromatin component M31. EMBO J. 1999;18:1923–1938. - PMC - PubMed
-
- Angelov D, Molla A, Perche PY, Hans F, Cote J, Khochbin S, Bouvet P, Dimitrov S. The histone variant macroH2A interferes with transcription factor binding and SWI/SNF nucleosome remodeling. Mol. Cell. 2003;11:1033–1041. - PubMed
-
- Bandyopadhyay D, Medrano EE. The emerging role of epigenetics in cellular and organismal aging. Exp. Gerontol. 2003;38:1299–1307. - PubMed
-
- Bandyopadhyay D, Mishra A, Medrano EE. Overexpression of histone deacetylase 1 confers resistance to sodium butyrate-mediated apoptosis in melanoma cells through a p53-mediated pathway. Cancer Res. 2004;64:7706–7710. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

