Size distribution of mouse Langerhans islets
- PMID: 17586568
- PMCID: PMC1989722
- DOI: 10.1529/biophysj.107.104125
Size distribution of mouse Langerhans islets
Abstract
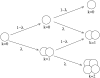
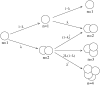
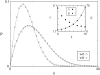
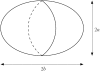
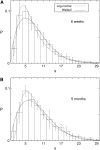
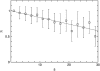
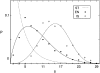
Pancreatic beta-cells are clustered in islets of Langerhans, which are typically a few hundred micrometers in a variety of mammals. In this study, we propose a theoretical model for the growth of pancreatic islets and derive the islet size distribution, based on two recent observations: First, the neogenesis of new islets becomes negligible after some developmental stage. Second, islets grow via a random process, where any cell in an islet proliferates with the same rate regardless of the present size of the islet. Our model predicts either log-normal or Weibull distributions of the islet sizes, depending on whether cells in an islet proliferate coherently or independently. To confirm this, we also measure the islet size by selectively staining islets, which are exposed from exocrine tissues in mice after enzymatic treatment. Indeed revealed are skewed distributions with the peak size of approximately 100 cells, which fit well to the theoretically derived ones. Interestingly, most islets turned out to be bigger than the expected minimal size (approximately 10 or so cells) necessary for stable synchronization of beta-cells through electrical gap-junction coupling. The collaborative behavior among cells is known to facilitate synchronized insulin secretion and tends to saturate beyond the critical (saturation) size of approximately 100 cells. We further probe how the islets change as normal mice grow from young (6 weeks) to adult (5 months) stages. It is found that islets may not grow too large to maintain appropriate ratios between cells of different types. Our results implicate that growing of mouse islets may be regulated by several physical constraints such as the minimal size required for stable cell-to-cell coupling and the upper limit to keep the ratios between cell types. Within the lower and upper limits the observed size distributions of islets can be faithfully regenerated by assuming random and uncoordinated proliferation of each beta-cell at appropriate rates.
Figures

References
-
- Brissova, M., M. J. Fowler, W. E. Nicholson, A. Chu, B. Hirshberg, D. M. Harlan, and A. C. Powers. 2005. Assessment of human pancreatic islet architecture and composition by laser scanning confocal microscopy. J. Histochem. Cytochem. 53:1087–1097. - PubMed
-
- Mears, D., N. F. Sheppard, I. Atwater, and E. Rojas. 1995. Magnitude and modulation of pancreatic β-cell gap junction electrical conductance in situ. J. Membr. Biol. 146:163–176. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

