Neural interpretation of blood oxygenation level-dependent fMRI maps at submillimeter columnar resolution
- PMID: 17596437
- PMCID: PMC6672231
- DOI: 10.1523/JNEUROSCI.0445-07.2007
Neural interpretation of blood oxygenation level-dependent fMRI maps at submillimeter columnar resolution
Abstract
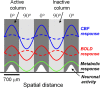
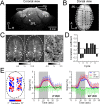
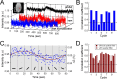
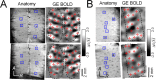
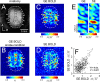
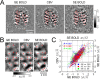
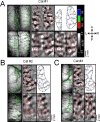
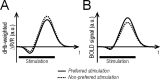
Whether conventional gradient-echo (GE) blood oxygenation-level-dependent (BOLD) functional magnetic resonance imaging (fMRI) is able to map submillimeter-scale functional columns remains debatable mainly because of the spatially nonspecific large vessel contribution, poor sensitivity and reproducibility, and lack of independent evaluation. Furthermore, if the results from optical imaging of intrinsic signals are directly applicable, regions with the highest BOLD signals may indicate neurally inactive domains rather than active columns when multiple columns are activated. To examine these issues, we performed BOLD fMRI at a magnetic field of 9.4 tesla to map orientation-selective columns of isoflurane-anesthetized cats. We could not convincingly map orientation columns using conventional block-design stimulation and differential analysis method because of large fluctuations of signals. However, we successfully obtained GE BOLD iso-orientation maps with high reproducibility (r = 0.74) using temporally encoded continuous cyclic orientation stimulation with Fourier data analysis, which reduces orientation-nonselective signals such as draining artifacts and is less sensitive to signal fluctuations. We further reduced large vessel contribution using the improved spin-echo (SE) BOLD method but with overall decreased sensitivity. Both GE and SE BOLD iso-orientation maps excluding large pial vascular regions were significantly correlated to maps with a known neural interpretation, which were obtained in contrast agent-aided cerebral blood volume fMRI and total hemoglobin-based optical imaging of intrinsic signals at a hemoglobin iso-sbestic point (570 nm). These results suggest that, unlike the expectation from deoxyhemoglobin-based optical imaging studies, the highest BOLD signals are localized to the sites of increased neural activity when column-nonselective signals are suppressed.
Figures

Similar articles
-
Mapping iso-orientation columns by contrast agent-enhanced functional magnetic resonance imaging: reproducibility, specificity, and evaluation by optical imaging of intrinsic signal.J Neurosci. 2006 Nov 15;26(46):11821-32. doi: 10.1523/JNEUROSCI.3098-06.2006. J Neurosci. 2006. PMID: 17108155 Free PMC article.
-
Spatiotemporal dynamics of the BOLD fMRI signals: toward mapping submillimeter cortical columns using the early negative response.Magn Reson Med. 2000 Aug;44(2):231-42. doi: 10.1002/1522-2594(200008)44:2<231::aid-mrm10>3.0.co;2-t. Magn Reson Med. 2000. PMID: 10918322
-
High-resolution mapping of iso-orientation columns by fMRI.Nat Neurosci. 2000 Feb;3(2):164-9. doi: 10.1038/72109. Nat Neurosci. 2000. PMID: 10649572
-
Lessons from fMRI about mapping cortical columns.Neuroscientist. 2008 Jun;14(3):287-99. doi: 10.1177/1073858407309541. Epub 2007 Nov 7. Neuroscientist. 2008. PMID: 17989170 Free PMC article. Review.
-
Biophysical and physiological origins of blood oxygenation level-dependent fMRI signals.J Cereb Blood Flow Metab. 2012 Jul;32(7):1188-206. doi: 10.1038/jcbfm.2012.23. Epub 2012 Mar 7. J Cereb Blood Flow Metab. 2012. PMID: 22395207 Free PMC article. Review.
Cited by
-
Surface-based analysis methods for high-resolution functional magnetic resonance imaging.Graph Models. 2011 Nov;73(6):313-322. doi: 10.1016/j.gmod.2010.11.002. Graph Models. 2011. PMID: 22125419 Free PMC article.
-
Modular head-mounted cortical imaging device for chronic monitoring of intrinsic signals in mice.J Biomed Opt. 2022 Feb;27(2):026501. doi: 10.1117/1.JBO.27.2.026501. J Biomed Opt. 2022. PMID: 35166087 Free PMC article.
-
Foundations of layer-specific fMRI and investigations of neurophysiological activity in the laminarized neocortex and olfactory bulb of animal models.Neuroimage. 2019 Oct 1;199:718-729. doi: 10.1016/j.neuroimage.2017.05.023. Epub 2017 May 12. Neuroimage. 2019. PMID: 28502845 Free PMC article. Review.
-
Time-dependent spatial specificity of high-resolution fMRI: insights into mesoscopic neurovascular coupling.Philos Trans R Soc Lond B Biol Sci. 2021 Jan 4;376(1815):20190623. doi: 10.1098/rstb.2019.0623. Epub 2020 Nov 16. Philos Trans R Soc Lond B Biol Sci. 2021. PMID: 33190606 Free PMC article. Review.
-
Demonstration of tuning to stimulus orientation in the human visual cortex: a high-resolution fMRI study with a novel continuous and periodic stimulation paradigm.Cereb Cortex. 2013 Jul;23(7):1618-29. doi: 10.1093/cercor/bhs149. Epub 2012 Jun 1. Cereb Cortex. 2013. PMID: 22661413 Free PMC article.
References
-
- Blasdel GG, Salama G. Voltage-sensitive dyes reveal a modular organization in monkey striate cortex. Nature. 1986;321:579–585. - PubMed
-
- Bosking WH, Crowley JC, Fitzpatrick D. Spatial coding of position and orientation in primary visual cortex. Nat Neurosci. 2002;5:874–882. - PubMed
-
- Cheng K, Waggoner RA, Tanaka K. Human ocular dominance columns as revealed by high-field functional magnetic resonance imaging. Neuron. 2001;32:359–374. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
