The acidic repetitive domain of the Magnetospirillum gryphiswaldense MamJ protein displays hypervariability but is not required for magnetosome chain assembly
- PMID: 17601786
- PMCID: PMC1951895
- DOI: 10.1128/JB.00421-07
The acidic repetitive domain of the Magnetospirillum gryphiswaldense MamJ protein displays hypervariability but is not required for magnetosome chain assembly
Abstract
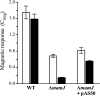
Magnetotactic bacteria navigate along the earth's magnetic field using chains of magnetosomes, which are intracellular organelles comprising membrane-enclosed magnetite crystals. The assembly of highly ordered magnetosome chains is under genetic control and involves several specific proteins. Based on genetic and cryo-electron tomography studies, a model was recently proposed in which the acidic MamJ magnetosome protein attaches magnetosome vesicles to the actin-like cytoskeletal filament formed by MamK, thereby preventing magnetosome chains from collapsing. However, the exact functions as well as the mode of interaction between MamK and MamJ are unknown. Here, we demonstrate that several functional MamJ variants from Magnetospirillum gryphiswaldense and other magnetotactic bacteria share an acidic and repetitive central domain, which displays an unusual intra- and interspecies sequence polymorphism, probably caused by homologous recombination between identical copies of Glu- and Pro-rich repeats. Surprisingly, mamJ mutant alleles in which the central domain was deleted retained their potential to restore chain formation in a DeltamamJ mutant, suggesting that the acidic domain is not essential for MamJ's function. Results of two-hybrid experiments indicate that MamJ physically interacts with MamK, and two distinct sequence regions within MamJ were shown to be involved in binding to MamK. Mutant variants of MamJ lacking either of the binding domains were unable to functionally complement the DeltamamJ mutant. In addition, two-hybrid experiments suggest both MamK-binding domains of MamJ confer oligomerization of MamJ. In summary, our data reveal domains required for the functions of the MamJ protein in chain assembly and maintenance and provide the first experimental indications for a direct interaction between MamJ and the cytoskeletal filament protein MamK.
Figures





References
-
- Ball, L. J., R. Kuhne, J. Schneider-Mergener, and H. Oschkinat. 2005. Recognition of proline-rich motifs by protein-protein-interaction domains. Angew. Chem. Int. Ed. 44:2852-2869. - PubMed
-
- Blakemore, R. P. 1975. Magnetotactic bacteria. Science 190:377-379. - PubMed
-
- Braun, V. 1995. Energy-coupled transport and signal-transduction through the Gram-negative outer-membrane via TonB-ExbB-ExbD-dependent receptor proteins. FEMS Microbiol. Rev. 16:295-307. - PubMed
-
- Dove, S. L., J. K. Joung, and A. Hochschild. 1997. Activation of prokaryotic transcription through arbitrary protein-protein contacts. Nature 386:627-630. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

