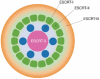
A concentric circle model of multivesicular body cargo sorting
- PMID: 17603537
- PMCID: PMC1905901
- DOI: 10.1038/sj.embor.7401004
A concentric circle model of multivesicular body cargo sorting
Abstract
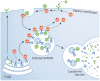
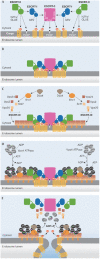
Targeting of ubiquitylated transmembrane proteins into luminal vesicles of endosomal multivesicular bodies (MVBs) depends on their recognition by endosomal sorting complexes required for transport (ESCRTs), which are also required for MVB vesicle formation. The model originally proposed for how ESCRTs function succinctly summarizes much of the protein-protein interaction and genetic data but oversimplifies the coordination of cargo recognition and cannot explain why ESCRTs are required for the budding of MVB vesicles. Recent structural and functional studies of ESCRT complexes suggest an alternative model that might direct the next series of breakthroughs in understanding protein sorting through the MVB pathway.
Figures




References
-
- Babst M (2005) A protein's final ESCRT. Traffic 6: 2–9 - PubMed
-
- Babst M, Katzmann DJ, Estepa-Sabal EJ, Meerloo T, Emr SD (2002a) ESCRT-III: an endosome-associated heterooligomeric protein complex required for MVB sorting. Dev Cell 3: 271–282 - PubMed
-
- Babst M, Katzmann DJ, Snyder WB, Wendland B, Emr SD (2002b) Endosome-associated complex, ESCRT-II, recruits transport machinery for protein sorting at the multivesicular body. Dev Cell 3: 283–289 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

