Mapping phosphoproteins in Mycoplasma genitalium and Mycoplasma pneumoniae
- PMID: 17605819
- PMCID: PMC1947986
- DOI: 10.1186/1471-2180-7-63
Mapping phosphoproteins in Mycoplasma genitalium and Mycoplasma pneumoniae
Abstract
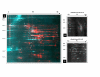
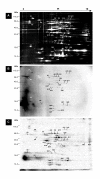
Background: Little is known regarding the extent or targets of phosphorylation in mycoplasmas, yet in many other bacterial species phosphorylation is known to play an important role in signaling and regulation of cellular processes. To determine the prevalence of phosphorylation in mycoplasmas, we examined the CHAPS-soluble protein fractions of Mycoplasma genitalium and Mycoplasma pneumoniae by two-dimensional gel electrophoresis (2-DE), using a combination of Pro-Q Diamond phosphoprotein stain and 33P labeling. Protein spots that were positive for phosphorylation were identified by peptide mass fingerprinting using MALDI-TOF-TOF mass spectrometry.
Results: We identified a total of 24 distinct phosphoproteins, about 3% and 5% of the total protein complement in M. pneumoniae and M. genitalium, respectively, indicating that phosphorylation occurs with prevalence similar to many other bacterial species. Identified phosphoproteins include pyruvate dehydrogenase E1 alpha and beta subunits, enolase, heat shock proteins DnaK and GroEL, elongation factor Tu, cytadherence accessory protein HMW3, P65, and several hypothetical proteins. These proteins are involved in energy metabolism, carbohydrate metabolism, translation/transcription and cytadherence. Interestingly, fourteen of the 24 phosphoproteins we identified (58%) were previously reported as putatively associated with a cytoskeleton-like structure that is present in the mycoplasmas, indicating a potential regulatory role for phosphorylation in this structure.
Conclusion: This study has shown that phosphorylation in mycoplasmas is comparable to that of other bacterial species. Our evidence supports a link between phosphorylation and cytadherence and/or a cytoskeleton-like structure, since over half of the proteins identified as phosphorylated have been previously associated with these functions. This opens the door to further research into the purposes and mechanisms of phosphorylation for mycoplasmas.
Figures

Similar articles
-
Elongation factor Tu and E1 beta subunit of pyruvate dehydrogenase complex act as fibronectin binding proteins in Mycoplasma pneumoniae.Mol Microbiol. 2002 Nov;46(4):1041-51. doi: 10.1046/j.1365-2958.2002.03207.x. Mol Microbiol. 2002. PMID: 12421310
-
Mycoplasma hyopneumoniae evades complement activation by binding to factor H via elongation factor thermo unstable (EF-Tu).Virulence. 2020 Dec;11(1):1059-1074. doi: 10.1080/21505594.2020.1806664. Virulence. 2020. PMID: 32815770 Free PMC article.
-
Amino acid changes in elongation factor Tu of Mycoplasma pneumoniae and Mycoplasma genitalium influence fibronectin binding.Infect Immun. 2009 Sep;77(9):3533-41. doi: 10.1128/IAI.00081-09. Epub 2009 Jun 22. Infect Immun. 2009. PMID: 19546194 Free PMC article.
-
Interplay between mycoplasma surface proteins, airway cells, and the protean manifestations of mycoplasma-mediated human infections.Am J Respir Crit Care Med. 1996 Oct;154(4 Pt 2):S137-44. doi: 10.1164/ajrccm/154.4_Pt_2.S137. Am J Respir Crit Care Med. 1996. PMID: 8876532 Review.
-
The mechanisms underlying antigenic variation and maintenance of genomic integrity in Mycoplasma pneumoniae and Mycoplasma genitalium.Arch Microbiol. 2021 Mar;203(2):413-429. doi: 10.1007/s00203-020-02041-4. Epub 2020 Sep 24. Arch Microbiol. 2021. PMID: 32970220 Review.
Cited by
-
Identification of novel immunogenic proteins from Mycoplasma bovis and establishment of an indirect ELISA based on recombinant E1 beta subunit of the pyruvate dehydrogenase complex.PLoS One. 2014 Feb 10;9(2):e88328. doi: 10.1371/journal.pone.0088328. eCollection 2014. PLoS One. 2014. PMID: 24520369 Free PMC article.
-
Why and how to investigate the role of protein phosphorylation in ZIP and ZnT zinc transporter activity and regulation.Cell Mol Life Sci. 2020 Aug;77(16):3085-3102. doi: 10.1007/s00018-020-03473-3. Epub 2020 Feb 19. Cell Mol Life Sci. 2020. PMID: 32076742 Free PMC article. Review.
-
Proteome characterization of developing grains in bread wheat cultivars (Triticum aestivum L.).BMC Plant Biol. 2012 Aug 19;12:147. doi: 10.1186/1471-2229-12-147. BMC Plant Biol. 2012. PMID: 22900893 Free PMC article.
-
Application of Proteomics Technologies in Oil Palm Research.Protein J. 2018 Dec;37(6):473-499. doi: 10.1007/s10930-018-9802-x. Protein J. 2018. PMID: 30367348 Review.
-
Complete genome and proteome of Acholeplasma laidlawii.J Bacteriol. 2011 Sep;193(18):4943-53. doi: 10.1128/JB.05059-11. Epub 2011 Jul 22. J Bacteriol. 2011. PMID: 21784942 Free PMC article.
References
-
- Fraser CM, Gocayne JD, White O, Adams MD, Clayton RA, Fleischmann RD, Bult CJ, Kerlavage AR, Sutton G, Kelley JM, Fritchman RD, Weidman JF, Small KV, Sandusky M, Fuhrmann J, Nguyen D, Utterback TR, Saudek DM, Phillips CA, Merrick JM, Tomb JF, Dougherty BA, Bott KF, Hu PC, Lucier TS, Peterson SN, Smith HO, Hutchison CA, Venter JC. The minimal gene complement of Mycoplasma genitalium. Science. 1995;270:397–403. - PubMed
-
- Hutchison CA, Peterson SN, Gill SR, Cline RT, White O, Fraser CM, Smith HO, Venter JC. Global transposon mutagenesis and a minimal mycoplasma genome. Science. 1999;286:2165–2169. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

