Cell division is essential for elimination of the yeast [PSI+] prion by guanidine hydrochloride
- PMID: 17606924
- PMCID: PMC1913874
- DOI: 10.1073/pnas.0701392104
Cell division is essential for elimination of the yeast [PSI+] prion by guanidine hydrochloride
Abstract
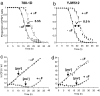
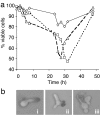
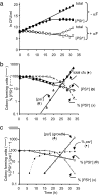
Guanidine hydrochloride (Gdn.HCl) blocks the propagation of yeast prions by inhibiting Hsp104, a molecular chaperone that is absolutely required for yeast prion propagation. We had previously proposed that ongoing cell division is required for Gdn.HCl-induced loss of the [PSI+] prion. Subsequently, Wu et al.[Wu Y, Greene LE, Masison DC, Eisenberg E (2005) Proc Natl Acad Sci USA 102:12789-12794] claimed to show that Gdn.HCl can eliminate the [PSI+] prion from alpha-factor-arrested cells leading them to propose that in Gdn.HCl-treated cells the prion aggregates are degraded by an Hsp104-independent mechanism. Here we demonstrate that the results of Wu et al. can be explained by an unusually high rate of alpha-factor-induced cell death in the [PSI+] strain (780-1D) used in their studies. What appeared to be no growth in their experiments was actually no increase in total cell number in a dividing culture through a counterbalancing level of cell death. Using media-exchange experiments, we provide further support for our original proposal that elimination of the [PSI+] prion by Gdn.HCl requires ongoing cell division and that prions are not destroyed during or after the evident curing phase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Guanidine hydrochloride inhibits the generation of prion "seeds" but not prion protein aggregation in yeast.Mol Cell Biol. 2002 Aug;22(15):5593-605. doi: 10.1128/MCB.22.15.5593-5605.2002. Mol Cell Biol. 2002. PMID: 12101251 Free PMC article.
-
Curing of yeast [PSI+] prion by guanidine inactivation of Hsp104 does not require cell division.Proc Natl Acad Sci U S A. 2005 Sep 6;102(36):12789-94. doi: 10.1073/pnas.0506384102. Epub 2005 Aug 25. Proc Natl Acad Sci U S A. 2005. PMID: 16123122 Free PMC article.
-
Guanidine hydrochloride inhibits Hsp104 activity in vivo: a possible explanation for its effect in curing yeast prions.Curr Microbiol. 2001 Jul;43(1):7-10. doi: 10.1007/s002840010251. Curr Microbiol. 2001. PMID: 11375656
-
Differential effects of chaperones on yeast prions: CURrent view.Curr Genet. 2018 Apr;64(2):317-325. doi: 10.1007/s00294-017-0750-3. Epub 2017 Sep 20. Curr Genet. 2018. PMID: 28932898 Review.
-
The story of stolen chaperones: how overexpression of Q/N proteins cures yeast prions.Prion. 2013 Jul-Aug;7(4):294-300. doi: 10.4161/pri.26021. Epub 2013 Aug 7. Prion. 2013. PMID: 23924684 Free PMC article. Review.
Cited by
-
Differences in the curing of [PSI+] prion by various methods of Hsp104 inactivation.PLoS One. 2012;7(6):e37692. doi: 10.1371/journal.pone.0037692. Epub 2012 Jun 18. PLoS One. 2012. PMID: 22719845 Free PMC article.
-
MIL-CELL: a tool for multi-scale simulation of yeast replication and prion transmission.Eur Biophys J. 2023 Nov;52(8):673-704. doi: 10.1007/s00249-023-01679-4. Epub 2023 Sep 5. Eur Biophys J. 2023. PMID: 37670150 Free PMC article.
-
Natural Genetic Variation in Yeast Reveals That NEDD4 Is a Conserved Modifier of Mutant Polyglutamine Aggregation.G3 (Bethesda). 2018 Nov 6;8(11):3421-3431. doi: 10.1534/g3.118.200289. G3 (Bethesda). 2018. PMID: 30194090 Free PMC article.
-
Osmolytes ameliorate the effects of stress in the absence of the heat shock protein Hsp104 in Saccharomyces cerevisiae.PLoS One. 2019 Sep 19;14(9):e0222723. doi: 10.1371/journal.pone.0222723. eCollection 2019. PLoS One. 2019. PMID: 31536559 Free PMC article.
-
Specificity of the J-protein Sis1 in the propagation of 3 yeast prions.Proc Natl Acad Sci U S A. 2008 Oct 28;105(43):16596-601. doi: 10.1073/pnas.0808934105. Epub 2008 Oct 27. Proc Natl Acad Sci U S A. 2008. PMID: 18955697 Free PMC article.
References
-
- Prusiner SB, Scott MR, DeArmound SJ, Cohen FE. Cell. 1998;93:337–348. - PubMed
-
- Uptain SM, Lindquist S. Annu Rev Microbiol. 2002;56:703–741. - PubMed
-
- Wickner RB, Edskes HK, Ross ED, Pierce MM, Baxa U, Brachmann A, Shewmaker F. Annu Rev Genet. 2004;38:681–707. - PubMed
-
- Tuite MF, Cox BS. Methods. 2006;39:9–22. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

