An erythroid chaperone that facilitates folding of alpha-globin subunits for hemoglobin synthesis
- PMID: 17607360
- PMCID: PMC1904324
- DOI: 10.1172/JCI31664
An erythroid chaperone that facilitates folding of alpha-globin subunits for hemoglobin synthesis
Abstract
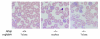
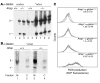
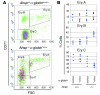
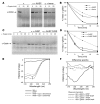
Erythrocyte precursors produce abundant alpha- and beta-globin proteins, which assemble with each other to form hemoglobin A (HbA), the major blood oxygen carrier. alphaHb-stabilizing protein (AHSP) binds free alpha subunits reversibly to maintain their structure and limit their ability to generate reactive oxygen species. Accordingly, loss of AHSP aggravates the toxicity of excessive free alpha-globin caused by beta-globin gene disruption in mice. Surprisingly, we found that AHSP also has important functions when free alpha-globin is limited. Thus, compound mutants lacking both Ahsp and 1 of 4 alpha-globin genes (genotype Ahsp(-/-)alpha-globin*(alpha/alphaalpha)) exhibited more severe anemia and Hb instability than mice with either mutation alone. In vitro, recombinant AHSP promoted folding of newly translated alpha-globin, enhanced its refolding after denaturation, and facilitated its incorporation into HbA. Moreover, in erythroid precursors, newly formed free alpha-globin was destabilized by loss of AHSP. Therefore, in addition to its previously defined role in detoxification of excess alpha-globin, AHSP also acts as a molecular chaperone to stabilize nascent alpha-globin for HbA assembly. Our findings illustrate what we believe to be a novel adaptive mechanism by which a specialized cell coordinates high-level production of a multisubunit protein and protects against various synthetic imbalances.
Figures


Comment in
-
AHSP: a novel hemoglobin helper.J Clin Invest. 2007 Jul;117(7):1746-9. doi: 10.1172/JCI32362. J Clin Invest. 2007. PMID: 17607349 Free PMC article.
Similar articles
-
The role of alpha-hemoglobin stabilizing protein in redox chemistry, denaturation, and hemoglobin assembly.Antioxid Redox Signal. 2010 Feb;12(2):219-31. doi: 10.1089/ars.2009.2780. Antioxid Redox Signal. 2010. PMID: 19659437 Free PMC article. Review.
-
Role of alpha-hemoglobin-stabilizing protein in normal erythropoiesis and beta-thalassemia.Ann N Y Acad Sci. 2005;1054:103-17. doi: 10.1196/annals.1345.013. Ann N Y Acad Sci. 2005. PMID: 16339656 Review.
-
Biochemical fates of alpha hemoglobin bound to alpha hemoglobin-stabilizing protein AHSP.J Biol Chem. 2006 Oct 27;281(43):32611-8. doi: 10.1074/jbc.M607311200. Epub 2006 Aug 10. J Biol Chem. 2006. PMID: 16901899
-
Transgenic human alpha-hemoglobin stabilizing protein could partially relieve betaIVS-2-654-thalassemia syndrome in model mice.Hum Gene Ther. 2010 Feb;21(2):149-56. doi: 10.1089/hum.2009.132. Hum Gene Ther. 2010. PMID: 20063986
-
Analysis of alpha hemoglobin stabilizing protein overexpression in murine β-thalassemia.Am J Hematol. 2010 Oct;85(10):820-2. doi: 10.1002/ajh.21829. Am J Hematol. 2010. PMID: 20815047 Free PMC article.
Cited by
-
The effect of histone deacetylase inhibitors on AHSP expression.PLoS One. 2018 Feb 1;13(2):e0189267. doi: 10.1371/journal.pone.0189267. eCollection 2018. PLoS One. 2018. PMID: 29389946 Free PMC article.
-
The role of alpha-hemoglobin stabilizing protein in redox chemistry, denaturation, and hemoglobin assembly.Antioxid Redox Signal. 2010 Feb;12(2):219-31. doi: 10.1089/ars.2009.2780. Antioxid Redox Signal. 2010. PMID: 19659437 Free PMC article. Review.
-
Role of α-globin H helix in the building of tetrameric human hemoglobin: interaction with α-hemoglobin stabilizing protein (AHSP) and heme molecule.PLoS One. 2014 Nov 4;9(11):e111395. doi: 10.1371/journal.pone.0111395. eCollection 2014. PLoS One. 2014. PMID: 25369055 Free PMC article.
-
Hemoglobin Variants as Targets for Stabilizing Drugs.Molecules. 2025 Jan 17;30(2):385. doi: 10.3390/molecules30020385. Molecules. 2025. PMID: 39860253 Free PMC article. Review.
-
A general map of iron metabolism and tissue-specific subnetworks.Mol Biosyst. 2009 May;5(5):422-43. doi: 10.1039/b816714c. Epub 2009 Mar 6. Mol Biosyst. 2009. PMID: 19381358 Free PMC article. Review.
References
-
- Dickerson, R.E., and Geis, I. 1983. Hemoglobin: structure, function, evolution, and pathology. Benjamin Cummings. Menlo Park, California, USA. 176 pp.
-
- Fermi, G., and Perutz, M.F. 1981. Atlas of molecular structures in biology. Volume 2, Haemoglobin and myoglobin. Oxford University Press. Oxford, United Kingdom. Clarendon Press. New York, New York, USA. 104 pp.
-
- Frydman J. Folding of newly translated proteins in vivo: the role of molecular chaperones. Annu. Rev. Biochem. 2001;70:603–647. - PubMed
-
- Forget, B.G., and Pearson, H.A. 1995. Hemoglobin synthesis and the thalassemias. In Blood: principles and practices of hematology. R.I. Handin, S.E. Lux, and T.P. Stossel, editors. J.B. Lippincott. Philadelphia, Pennsylvania, USA. 1525–1990.
-
- Nathan D.G., Gunn R.B. Thalassemia: the consequences of unbalanced hemoglobin synthesis. Am. J. Med. 1966;41:815–830. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

