Addition of TAT protein transduction domain and GrpE to human p53 provides soluble fusion proteins that can be transduced into dendritic cells and elicit p53-specific T-cell responses in HLA-A*0201 transgenic mice
- PMID: 17610503
- PMCID: PMC2266015
- DOI: 10.1111/j.1365-2567.2007.02643.x
Addition of TAT protein transduction domain and GrpE to human p53 provides soluble fusion proteins that can be transduced into dendritic cells and elicit p53-specific T-cell responses in HLA-A*0201 transgenic mice
Abstract
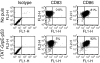
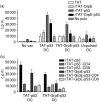
The protein p53 has been shown to be an efficient tumour antigen in both murine and human cancer vaccine studies and cancer vaccines targeting p53 based on major histocompatibility complex (MHC) class I binding p53-derived peptides that induce cytotoxic T lymphocytes (CTLs) without p53-specific CD4(+) T-cell help have been tested by several research groups including ours. To obtain such CD4(+) T-cell help and cover a broader repertoire of MHC haplotypes we have previously attempted to produce recombinant human p53 for vaccination purposes. However, attempts to refold a hexahis-tagged p53 protein in our laboratory were unsuccessful. Here, we show that fusion of an 11-amino-acid region of the human immunodeficiency virus TAT protein transduction domain (PTD) to human p53 increases the solubility of the otherwise insoluble p53 protein and this rTAT-p53 protein can be transduced into human monocyte-derived dendritic cells (DCs). The induction of a p53-specific HLA-A*0201 immune response was tested in HLA-A*0201/K(b) transgenic mice after immunization with rTAT-p53-transduced bone-marrow-derived DCs. In these mice, p53-specific CD4(+) and CD8(+) T-cell proliferation was observed and immunization resulted in the induction of HLA-A*0201-restricted CTLs specific for two human p53-derived HLA-A*0201-binding peptides, p53(65-73) and p53(149-157). Addition of GrpE to generate rTAT-GrpE-p53 led to a further increase in protein solubility and to a small increase in DC maturation but did not increase the observed p53-specific T-cell responses. The use of rTAT-p53 in ongoing clinical protocols should be applicable and offers advantages to current strategies omitting the use of HLA-typed patients.
Figures




References
-
- Guermonprez P, Valladeau J, Zitvogel L, Thery C, Amigorena S. Antigen presentation and T cell stimulation by dendritic cells. Annu Rev Immunol. 2002;20:621–67. - PubMed
-
- Figdor CGDE, Vries IJ, Lesterhuis WJ, Melief CJ. Dendritic cell immunotherapy: mapping the way. Nat Med. 2004;10:475–80. - PubMed
-
- Offringa R, Vierboom MP, van der Burg SH, Erdile L, Melief CJ. p53. a potential target antigen for immunotherapy of cancer. Ann N Y Acad Sci. 2000 June;910:223–33. - PubMed
-
- Soussi T. The p53 tumor suppressor gene: from molecular biology to clinical investigation. Ann N Y Acad Sci. 2000;910:121–37. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

