An increase in cell number at completion of therapy may develop as an indicator of early relapse: quantification of circulating epithelial tumor cells (CETC) for monitoring of adjuvant therapy in breast cancer
- PMID: 17611779
- PMCID: PMC12161744
- DOI: 10.1007/s00432-007-0248-3
An increase in cell number at completion of therapy may develop as an indicator of early relapse: quantification of circulating epithelial tumor cells (CETC) for monitoring of adjuvant therapy in breast cancer
Abstract
Purpose: Treatment efficiency of adjuvant therapy in breast cancer is only revealed after several years by statistical evaluation and gives no answer for the individual patient. We here present a method to analyze the response to adjuvant chemotherapy online in individual patients.
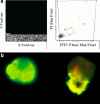
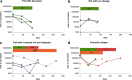
Methods/results: In 25 consecutive non-metastatic primary breast cancer patients adjuvant fluorouracil/epirubicin/cyclophosphamid (FEC) or EC followed by taxane (EC-T) or cyclophosphamid/methotrexate/fluorouracil (CMF) therapy were given. Circulating epithelial tumor cells (CETC) were quantified before and after each second cycle of the therapy regimen, between the anthracycline and the taxane block of the regimen and in some cases repeatedly during CMF treatment. Independent of the initial cell number CETC numbers showed a decline, no change or a minor increase in 15 patients of which 14 remained in complete remission and 1 suffered local relapse. Ten patients showed an increase at the end of therapy of which 4 have relapsed during the observation time of between 2 months and up to 54 months. This patient group was compared to a previously published group of 25 patients who have all reached a follow-up of 4.5 years or until relapse.
Conclusion: As in the previous report, Kaplan-Meier analysis revealed a high correlation between the response of CETC to therapy and relapse (p < 0.0001) and curves of both patient groups were super imposable. Multivariate analysis revealed the response of CETC to therapy to be an independent predictive marker for relapse.
Figures

Similar articles
-
WITHDRAWN: Multi-agent chemotherapy for early breast cancer.Cochrane Database Syst Rev. 2008 Oct 8;2008(4):CD000487. doi: 10.1002/14651858.CD000487.pub2. Cochrane Database Syst Rev. 2008. PMID: 18843612 Free PMC article.
-
Multi-agent chemotherapy for early breast cancer.Cochrane Database Syst Rev. 2002;(1):CD000487. doi: 10.1002/14651858.CD000487. Cochrane Database Syst Rev. 2002. Update in: Cochrane Database Syst Rev. 2008 Oct 08;(4):CD000487. doi: 10.1002/14651858.CD000487.pub2. PMID: 11869577 Updated.
-
Taxane monotherapy regimens for the treatment of recurrent epithelial ovarian cancer.Cochrane Database Syst Rev. 2022 Jul 12;7(7):CD008766. doi: 10.1002/14651858.CD008766.pub3. Cochrane Database Syst Rev. 2022. PMID: 35866378 Free PMC article.
-
Effectiveness of an Adjuvant Chemotherapy Regimen for Early-Stage Breast Cancer: A Systematic Review and Network Meta-analysis.JAMA Oncol. 2015 Dec;1(9):1311-8. doi: 10.1001/jamaoncol.2015.3062. JAMA Oncol. 2015. PMID: 26402167 Free PMC article.
-
A systematic overview of chemotherapy effects in breast cancer.Acta Oncol. 2001;40(2-3):253-81. doi: 10.1080/02841860151116349. Acta Oncol. 2001. PMID: 11441936
Cited by
-
Evaluation of a multi-marker immunomagnetic enrichment assay for the quantification of circulating melanoma cells.J Transl Med. 2012 Sep 15;10:192. doi: 10.1186/1479-5876-10-192. J Transl Med. 2012. PMID: 22978632 Free PMC article.
-
Treatment of advanced solid tumours with NSAIDs: Correlation of quantitative monitoring of circulating tumour cells and positron emission tomography-computed tomography imaging.Oncol Lett. 2016 Sep;12(3):1711-1716. doi: 10.3892/ol.2016.4878. Epub 2016 Jul 18. Oncol Lett. 2016. PMID: 27588120 Free PMC article.
-
Future directions for the early detection of recurrent breast cancer.J Cancer. 2014 Mar 16;5(4):291-300. doi: 10.7150/jca.8017. eCollection 2014. J Cancer. 2014. PMID: 24790657 Free PMC article. Review.
-
Increased Circulating Epithelial Tumor Cells (CETC/CTC) over the Course of Adjuvant Radiotherapy Is a Predictor of Less Favorable Outcome in Patients with Early-Stage Breast Cancer.Curr Oncol. 2022 Dec 24;30(1):261-273. doi: 10.3390/curroncol30010021. Curr Oncol. 2022. PMID: 36661670 Free PMC article. Clinical Trial.
-
High depletion of breast cancer cells from the peripheral blood with the method of non-specific separation.Ecancermedicalscience. 2020 Jan 21;14:1003. doi: 10.3332/ecancer.2020.1003. eCollection 2020. Ecancermedicalscience. 2020. PMID: 32104205 Free PMC article.
References
-
- Braun S, Vogl FD, Naume B, Janni W, Osborne MP, Coombes RC, Schlimok G, Diel IJ, Gerber B, Gebauer G, Pierga JY, Marth C, Oruzio D, Wiedswang G, Solomayer EF, Kundt G, Strobl B, Fehm T, Wong GY, Bliss J, Vincent-Salomon A, Pantel K (2005) A pooled analysis of bone marrow micrometastasis in breast cancer. N Engl J Med 353:793–802 - PubMed
-
- Campone M, Fumoleau P, Bourbouloux E, Kerbrat P, Roche H (2005) Taxanes in adjuvant breast cancer setting: which standard in Europe? Crit Rev Oncol Hematol 55:167–175 - PubMed
-
- Carrick S, Parker S, Wilcken N, Ghersi D, Marzo M, Simes J (2005) Single agent versus combination chemotherapy for metastatic breast cancer. Cochrane Database Syst Rev. Apr 18:CD003372 - PubMed
-
- Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ, Terstappen LW, Hayes DF (2004) Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351:781–791 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

