Synthesis and biological evaluation of phosphate prodrugs of 4-phospho-D-erythronohydroxamic acid, an inhibitor of 6-phosphogluconate dehydrogenase
- PMID: 17615587
- PMCID: PMC2248282
- DOI: 10.1002/cmdc.200700040
Synthesis and biological evaluation of phosphate prodrugs of 4-phospho-D-erythronohydroxamic acid, an inhibitor of 6-phosphogluconate dehydrogenase
Abstract
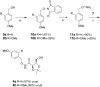
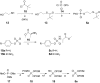
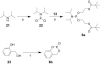
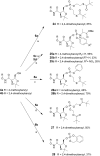
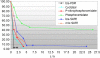
We have previously reported the discovery of potent and selective inhibitors of 6-phosphogluconate dehydrogenase, the third enzyme of the phosphate pentose pathway, from Trypanosoma brucei, the causative organism of human African trypanosomiasis. These inhibitors were charged phosphate derivatives with restricted capacity to enter cells. Herein, we report the synthesis of five different classes of prodrugs: phosphoramidate; bis-S-acyl thioethyl esters (bis-SATE); bis-pivaloxymethyl (bis-POM); CycloSaligenyl; and phenyl, S-acyl thioethyl mixed phosphate esters (mix-SATE). Prodrugs were studied for stability and activity against the intact parasites. Most prodrugs caused inhibition of the growth of the parasites. The activity of the prodrugs against the parasites appeared to be related to their stability in aqueous buffer.
Figures
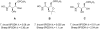
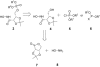
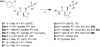
Similar articles
-
Aryl phosphoramidates of 5-phospho erythronohydroxamic acid, a new class of potent trypanocidal compounds.J Med Chem. 2010 Aug 26;53(16):6071-8. doi: 10.1021/jm1004754. J Med Chem. 2010. PMID: 20666371 Free PMC article.
-
Thermodynamic characterization of substrate and inhibitor binding to Trypanosoma brucei 6-phosphogluconate dehydrogenase.FEBS J. 2007 Dec;274(24):6426-35. doi: 10.1111/j.1742-4658.2007.06160.x. Epub 2007 Nov 15. FEBS J. 2007. PMID: 18021252
-
6-phosphogluconate dehydrogenase: a target for drugs in African trypanosomes.Curr Med Chem. 2004 Oct;11(19):2639-50. doi: 10.2174/0929867043364441. Curr Med Chem. 2004. PMID: 15544466 Review.
-
Selective inhibition of Trypanosoma brucei 6-phosphogluconate dehydrogenase by high-energy intermediate and transition-state analogues.J Med Chem. 2004 Jun 17;47(13):3427-37. doi: 10.1021/jm031066i. J Med Chem. 2004. PMID: 15189039
-
Novel approaches for designing 5'-O-ester prodrugs of 3'-azido-2', 3'-dideoxythymidine (AZT).Curr Med Chem. 2000 Oct;7(10):995-1039. doi: 10.2174/0929867003374372. Curr Med Chem. 2000. PMID: 10911016 Review.
Cited by
-
Phosphoramidates and phosphonamidates (ProTides) with antiviral activity.Antivir Chem Chemother. 2018 Jan-Dec;26:2040206618775243. doi: 10.1177/2040206618775243. Antivir Chem Chemother. 2018. PMID: 29792071 Free PMC article. Review.
-
Essential multimeric enzymes in kinetoplastid parasites: A host of potentially druggable protein-protein interactions.PLoS Negl Trop Dis. 2017 Jun 29;11(6):e0005720. doi: 10.1371/journal.pntd.0005720. eCollection 2017 Jun. PLoS Negl Trop Dis. 2017. PMID: 28662026 Free PMC article.
-
Virtual fragment screening for novel inhibitors of 6-phosphogluconate dehydrogenase.Bioorg Med Chem. 2010 Jul 15;18(14):5056-62. doi: 10.1016/j.bmc.2010.05.077. Epub 2010 Jun 9. Bioorg Med Chem. 2010. PMID: 20598892 Free PMC article.
-
Enzyme-Catalyzed Kinetic Resolution of Chiral Precursors to Antiviral Prodrugs.Biochemistry. 2019 Jul 23;58(29):3204-3211. doi: 10.1021/acs.biochem.9b00530. Epub 2019 Jul 10. Biochemistry. 2019. PMID: 31268686 Free PMC article.
-
The immune response to RNA suppresses nucleic acid synthesis by limiting ribose 5-phosphate.EMBO J. 2024 Jul;43(13):2636-2660. doi: 10.1038/s44318-024-00100-w. Epub 2024 May 22. EMBO J. 2024. PMID: 38778156 Free PMC article.
References
-
- “Human African trypanosomiasis”. to be found under http://www.who.int/tdr/diseases/tryp/diseaseinfo.htm.
-
- WHO Fact sheet N°259—“African trypanosomiasis (sleeping sickness)”. to be found under http://www.who.int/mediacentre/factsheets/fs259/en/
-
- Opperdoes FR, Michels PAM. Int. J. Parasitol. 2001;31:482. - PubMed
-
- Hanau S, Rinaldi E, Dallocchio F, Gilbert IH, Dardonville C, Adams MJ, Gover S, Barrett MP. Curr. Med. Chem. 2004;11:2639. - PubMed
-
- Barrett MP. Parasitol. Today. 1997;13:11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

