Maternal transmission of resistance to development of allergic airway disease
- PMID: 17617621
- PMCID: PMC3155847
- DOI: 10.4049/jimmunol.179.2.1282
Maternal transmission of resistance to development of allergic airway disease
Abstract
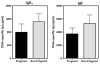
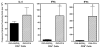
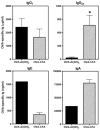
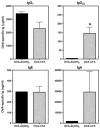
Parental phenotype is known to influence the inheritance of atopic diseases, such as allergic asthma, with a maternal history being a more significant risk factor for progeny than paternal history. We hypothesized that recall Th1- or Th2-type immune responses during pregnancy would result in transfer of maternal factors that would differentially impact development of immune responsiveness in offspring. Following weaning, susceptibility and severity of allergic airway disease (a murine model of human asthma) was evaluated in progeny, disease being elicited by immunization with OVA-Al(OH)(3) and challenge with aerosolized OVA. We found that progeny of mothers with Th1-biased immunity to OVA subjected to recall aerosol challenge during pregnancy had reduced levels of Ag-specific IgE and airway eosinophilia compared with progeny of mothers with Th2-biased immunity to OVA or naive mothers. Interestingly, progeny of mothers with Th1-type immunity to a heterologous albumin, BSA, were not protected from developing OVA-induced allergic airway disease. These findings demonstrated that maternal transfer of protection from development of allergic airway disease to offspring in this model of maternal Th1-type immunity was Ag specific.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures


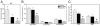
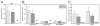
Similar articles
-
Maternal allergen immunisation to prevent sensitisation in offspring: Th2-polarising adjuvants are more efficient than a Th1-polarising adjuvant in mice.BMC Immunol. 2010 Mar 1;11:8. doi: 10.1186/1471-2172-11-8. BMC Immunol. 2010. PMID: 20193059 Free PMC article.
-
Breastmilk from allergic mothers can protect offspring from allergic airway inflammation.Breastfeed Med. 2009 Sep;4(3):167-74. doi: 10.1089/bfm.2008.0130. Breastfeed Med. 2009. PMID: 19301986 Free PMC article.
-
Modulation of Th2 responses by peptide analogues in a murine model of allergic asthma: amelioration or deterioration of the disease process depends on the Th1 or Th2 skewing characteristics of the therapeutic peptide.J Immunol. 2000 Jan 15;164(2):580-8. doi: 10.4049/jimmunol.164.2.580. J Immunol. 2000. PMID: 10623798
-
[A role for T-helper type 1 and type 2 cytokines in the pathogenesis of various human diseases].Rinsho Byori. 1998 Sep;46(9):915-21. Rinsho Byori. 1998. PMID: 9800477 Review. Japanese.
-
Maternal influence in the transmission of asthma susceptibility.Pulm Pharmacol Ther. 2008;21(3):474-84. doi: 10.1016/j.pupt.2007.06.005. Epub 2007 Jul 12. Pulm Pharmacol Ther. 2008. PMID: 17693106 Free PMC article. Review.
Cited by
-
Fetal-maternal alignment of regulatory T cells correlates with IL-10 and Bcl-2 upregulation in pregnancy.J Immunol. 2013 Jul 1;191(1):145-53. doi: 10.4049/jimmunol.1203165. Epub 2013 Jun 3. J Immunol. 2013. PMID: 23733877 Free PMC article.
-
Mucosal immunology of food allergy.Curr Biol. 2013 May 6;23(9):R389-400. doi: 10.1016/j.cub.2013.02.043. Curr Biol. 2013. PMID: 23660362 Free PMC article. Review.
-
Mucosal antibodies in the regulation of tolerance and allergy to foods.Semin Immunopathol. 2012 Sep;34(5):633-42. doi: 10.1007/s00281-012-0325-9. Epub 2012 Jul 10. Semin Immunopathol. 2012. PMID: 22777546 Free PMC article. Review.
-
The Potential of IgG to Induce Murine and Human Thymic Maturation of IL-10+ B Cells (B10) Revealed in a Pilot Study.Cells. 2020 Oct 5;9(10):2239. doi: 10.3390/cells9102239. Cells. 2020. PMID: 33027887 Free PMC article.
-
Preclinical models of maternal asthma and progeny outcomes: a scoping review.Eur Respir Rev. 2024 Feb 28;33(171):230174. doi: 10.1183/16000617.0174-2023. Print 2024 Jan 31. Eur Respir Rev. 2024. PMID: 38417970 Free PMC article.
References
-
- Walker C, Kaegi MK, Braun P, Blaser K. Activated T cells and eosinophilia in bronchoalveolar lavages from subjects with asthma correlated with disease severity. J Allergy Clin Immunol. 1991;88:935–942. - PubMed
-
- McFadden ER, I, Gilbert A. Asthma. N Engl J Med. 1992;327:1928–1924. - PubMed
-
- Corrigan CJ, Kay AB. Asthma: role of T-lymphocytes and lymphokines. Br Med Bull. 1992;48:72– 84. - PubMed
-
- Corrigan CJ, Kay AB. T cells and eosinophils in the pathogenesis of asthma. Immunol Today. 1992;13:501–507. - PubMed
-
- Wilson JW, Djukanovic R, Howarth PH, Holgate ST. Lymphocyte activation in bronchoalveolar lavage and peripheral blood in atopic asthma. Am Rev Respir Dis. 1992;145:958–960. - PubMed

