Mutations in the Drosophila alphaPS2 integrin subunit uncover new features of adhesion site assembly
- PMID: 17618618
- PMCID: PMC3861690
- DOI: 10.1016/j.ydbio.2007.02.046
Mutations in the Drosophila alphaPS2 integrin subunit uncover new features of adhesion site assembly
Abstract
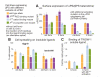
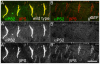
The Drosophila alphaPS2betaPS integrin is required for diverse development events, including muscle attachment. We characterized six unusual mutations in the alphaPS2 gene that cause a subset of the null phenotype. One mutation changes a residue in alphaPS2 that is equivalent to the residue in alphaV that contacts the arginine of RGD. This change severely reduced alphaPS2betaPS affinity for soluble ligand, abolished the ability of the integrin to recruit laminin to muscle attachment sites in the embryo and caused detachment of integrins and talin from the ECM. Three mutations that alter different parts of the alphaPS2 beta-propeller, plus a fourth that eliminated a late phase of alphaPS2 expression, all led to a strong decrease in alphaPS2betaPS at muscle ends, but, surprisingly, normal levels of talin were recruited. Thus, although talin recruitment requires alphaPS2betaPS, talin levels are not simply specified by the amount of integrin at the adhesive junction. These mutations caused detachment of talin and actin from integrins, suggesting that the integrin-talin link is weaker than the ECM-integrin link.
Figures










Similar articles
-
Amino acid changes in Drosophila alphaPS2betaPS integrins that affect ligand affinity.J Biol Chem. 2006 Feb 24;281(8):5050-7. doi: 10.1074/jbc.M508550200. Epub 2005 Dec 21. J Biol Chem. 2006. PMID: 16371365
-
Distinct developmental roles for direct and indirect talin-mediated linkage to actin.Dev Biol. 2010 Sep 1;345(1):64-77. doi: 10.1016/j.ydbio.2010.06.027. Epub 2010 Jun 28. Dev Biol. 2010. PMID: 20599891
-
Requirements for the cytoplasmic domain of the alphaPS1, alphaPS2 and betaPS integrin subunits during Drosophila development.Development. 1998 Feb;125(4):701-11. doi: 10.1242/dev.125.4.701. Development. 1998. PMID: 9435290
-
Talin - the master of integrin adhesions.J Cell Sci. 2017 Aug 1;130(15):2435-2446. doi: 10.1242/jcs.190991. Epub 2017 Jul 12. J Cell Sci. 2017. PMID: 28701514 Review.
-
Integrin-mediated cell adhesion: the cytoskeletal connection.Biochem Soc Symp. 1999;65:79-99. Biochem Soc Symp. 1999. PMID: 10320934 Review.
Cited by
-
An O-glycosyltransferase promotes cell adhesion during development by influencing secretion of an extracellular matrix integrin ligand.J Biol Chem. 2010 Jun 18;285(25):19491-501. doi: 10.1074/jbc.M109.098145. Epub 2010 Apr 6. J Biol Chem. 2010. PMID: 20371600 Free PMC article.
-
RacGAP50C directs perinuclear gamma-tubulin localization to organize the uniform microtubule array required for Drosophila myotube extension.Development. 2009 May;136(9):1411-21. doi: 10.1242/dev.031823. Epub 2009 Mar 18. Development. 2009. PMID: 19297411 Free PMC article.
-
Differences in regulation of Drosophila and vertebrate integrin affinity by talin.Mol Biol Cell. 2008 Aug;19(8):3589-98. doi: 10.1091/mbc.e08-01-0085. Epub 2008 May 28. Mol Biol Cell. 2008. PMID: 18508915 Free PMC article.
-
Cell adhesion in Drosophila: versatility of cadherin and integrin complexes during development.Curr Opin Cell Biol. 2012 Oct;24(5):702-12. doi: 10.1016/j.ceb.2012.07.006. Epub 2012 Aug 28. Curr Opin Cell Biol. 2012. PMID: 22938782 Free PMC article. Review.
-
Parvin-ILK: An intimate relationship.Bioarchitecture. 2012 May 1;2(3):91-94. doi: 10.4161/bioa.20700. Bioarchitecture. 2012. PMID: 22880148 Free PMC article.
References
-
- Basani RB, Vilaire G, Shattil SJ, Kolodziej MA, Bennett JS, Poncz M. Glanzmann thrombasthenia due to a two amino acid deletion in the fourth calcium-binding domain of alpha IIb: demonstration of the importance of calcium-binding domains in the conformation of alpha IIb beta 3. Blood. 1996;88:167–73. - PubMed
-
- Bobinnec Y, Marcaillou C, Morin X, Debec A. Dynamics of the endoplasmic reticulum during early development of Drosophila melanogaster. Cell Motil Cytoskeleton. 2003;54:217–25. - PubMed
-
- Bokel C, Brown NH. Integrins in development: moving on, responding to, and sticking to the extracellular matrix. Dev Cell. 2002;3:311–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

