Characterization of thimet oligopeptidase and neurolysin activities in B16F10-Nex2 tumor cells and their involvement in angiogenesis and tumor growth
- PMID: 17620116
- PMCID: PMC1965469
- DOI: 10.1186/1476-4598-6-44
Characterization of thimet oligopeptidase and neurolysin activities in B16F10-Nex2 tumor cells and their involvement in angiogenesis and tumor growth
Abstract
Background: Angiogenesis is a fundamental process that allows tumor growth by providing nutrients and oxygen to the tumor cells. Beyond the oxygen diffusion limit from a capillary blood vessel, tumor cells become apoptotic. Angiogenesis results from a balance of pro- and anti-angiogenic stimuli. Endogenous inhibitors regulate enzyme activities that promote angiogenesis. Tumor cells may express pro-angiogenic factors and hydrolytic enzymes but also kinin-degrading oligopeptidases which have been investigated.
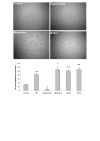
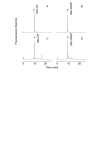
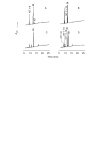
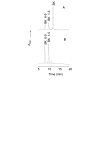
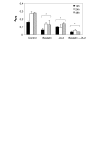
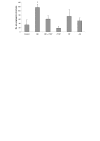
Results: Angiogenesis induced by B16F10-Nex2 melanoma cells was studied in a co-culture with HUVEC on Matrigel. A stimulating effect on angiogenesis was observed in the presence of B16F10-Nex2 lysate and plasma membrane. In contrast, the B16F10-Nex2 culture supernatant inhibited angiogenesis in a dose-dependent manner. This effect was abolished by the endo-oligopeptidase inhibitor, JA-2. Thimet oligopeptidase (TOP) and neurolysin activities were then investigated in B16F10-Nex2 melanoma cells aiming at gene sequencing, enzyme distribution and activity, influence on tumor development, substrate specificity, hydrolytic products and susceptibility to inhibitors. Fluorescence resonance energy transfer (FRET) peptides as well as neurotensin and bradykinin were used as substrates. The hydrolytic activities in B16F10-Nex2 culture supernatant were totally inhibited by o-phenanthrolin, JA-2 and partially by Pro-Ile. Leupeptin, PMSF, E-64, Z-Pro-Prolinal and captopril failed to inhibit these hydrolytic activities. Genes encoding M3A enzymes in melanoma cells were cloned and sequenced being highly similar to mouse genes. A decreased proliferation of B16F10-Nex2 cells was observed in vitro with specific inhibitors of these oligopeptidases. Active rTOP but not the inactive protein inhibited melanoma cell development in vivo increasing significantly the survival of mice challenged with the tumor cells. On Matrigel, rTOP inhibited the bradykinin - induced angiogenesis. A possible regulation of the homologous tumor enzyme in the perivascular microenvironment is suggested based on the observed rTOP inhibition by an S-nitrosothiol NO donor.
Conclusion: Data show that melanoma cells secrete endo-oligopeptidases which have an important role in tumor proliferation in vitro and in vivo. rTOP inhibited growth of subcutaneously injected B16F10-Nex2 cells in mice. TOP from tumor cells and bradykinin in endothelial cells are two antagonist factors that may control angiogenesis essential for melanoma growth. A regulatory role of NO or S-nitrosothiols is suggested.
Figures



Similar articles
-
Characterization of thimet- and neurolysin-like activities in Escherichia coli M 3 A peptidases and description of a specific substrate.Arch Biochem Biophys. 2005 Sep 1;441(1):25-34. doi: 10.1016/j.abb.2005.06.011. Arch Biochem Biophys. 2005. PMID: 16098472
-
Role of the SIKVAV site of laminin in promotion of angiogenesis and tumor growth: an in vivo Matrigel model.J Natl Cancer Inst. 1992 Nov 4;84(21):1633-8. doi: 10.1093/jnci/84.21.1633. J Natl Cancer Inst. 1992. PMID: 1279183
-
Danshensu has anti-tumor activity in B16F10 melanoma by inhibiting angiogenesis and tumor cell invasion.Eur J Pharmacol. 2010 Sep 25;643(2-3):195-201. doi: 10.1016/j.ejphar.2010.06.045. Epub 2010 Jul 11. Eur J Pharmacol. 2010. PMID: 20621088
-
Angiogenesis and cancer prevention: a vision.Recent Results Cancer Res. 2007;174:219-24. doi: 10.1007/978-3-540-37696-5_19. Recent Results Cancer Res. 2007. PMID: 17302199 Review.
-
An Overview of In Vitro, In Vivo, and Computational Techniques for Cancer-Associated Angiogenesis Studies.Biomed Res Int. 2020 Dec 10;2020:8857428. doi: 10.1155/2020/8857428. eCollection 2020. Biomed Res Int. 2020. PMID: 33381591 Free PMC article. Review.
Cited by
-
Serum From Advanced Heart Failure Patients Promotes Angiogenic Sprouting and Affects the Notch Pathway in Human Endothelial Cells.J Cell Physiol. 2016 Dec;231(12):2700-10. doi: 10.1002/jcp.25373. Epub 2016 Apr 4. J Cell Physiol. 2016. PMID: 26987674 Free PMC article.
-
Components in melanoma cytoplasm might induce murine BMSCs transformation and expression of Melan-A.J Huazhong Univ Sci Technolog Med Sci. 2011 Oct;31(5):663. doi: 10.1007/s11596-011-0578-x. Epub 2011 Oct 25. J Huazhong Univ Sci Technolog Med Sci. 2011. PMID: 22038357
-
Differential Antitumor Effects of IgG and IgM Monoclonal Antibodies and Their Synthetic Complementarity-Determining Regions Directed to New Targets of B16F10-Nex2 Melanoma Cells.Transl Oncol. 2010 Aug 1;3(4):204-17. doi: 10.1593/tlo.09316. Transl Oncol. 2010. PMID: 20689762 Free PMC article.
-
A novel melanoma-targeting peptide screened by phage display exhibits antitumor activity.J Mol Med (Berl). 2010 Dec;88(12):1255-64. doi: 10.1007/s00109-010-0671-9. Epub 2010 Aug 28. J Mol Med (Berl). 2010. PMID: 20802991
-
17β-estradiol enhances signalling mediated by VEGF-A-delta-like ligand 4-notch1 axis in human endothelial cells.PLoS One. 2013 Aug 13;8(8):e71440. doi: 10.1371/journal.pone.0071440. eCollection 2013. PLoS One. 2013. PMID: 23967210 Free PMC article.
References
-
- Freitas ZFO, Rodrigues EG, Oliveira V, Carmona AK, Travassos LR. Melanoma heterogeneity: differential, invasive, metastatic properties and profiles of cathepsin B, D and L activities in subclones of the B16F10-NEX2 cell line. Melanoma Res. 2004;14:333–344. doi: 10.1097/00008390-200410000-00002. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

