Influence of the Cpx extracytoplasmic-stress-responsive pathway on Yersinia sp.-eukaryotic cell contact
- PMID: 17620356
- PMCID: PMC1951158
- DOI: 10.1128/IAI.01450-06
Influence of the Cpx extracytoplasmic-stress-responsive pathway on Yersinia sp.-eukaryotic cell contact
Abstract
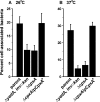
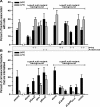
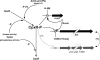
The extracytoplasmic-stress-responsive CpxRA two-component signal transduction pathway allows bacteria to adapt to growth in extreme environments. It controls the production of periplasmic protein folding and degradation factors, which aids in the biogenesis of multicomponent virulence determinants that span the bacterial envelope. This is true of the Yersinia pseudotuberculosis Ysc-Yop type III secretion system. However, despite using a second-site suppressor mutation to restore Yop effector secretion by yersiniae defective in the CpxA sensor kinase, these bacteria poorly translocated Yops into target eukaryotic cells. Investigation of this phenotype herein revealed that the expression of genes which encode several surface-located adhesins is also influenced by the Cpx pathway. In particular, the expression and surface localization of invasin, an adhesin that engages beta1-integrins on the eukaryotic cell surface, are severely restricted by the removal of CpxA. This reduces bacterial association with eukaryotic cells, which could be suppressed by the ectopic production of CpxA, invasin, or RovA, a positive activator of inv expression. In turn, these infected eukaryotic cells then became susceptible to intoxication by translocated Yop effectors. In contrast, bacteria harboring an in-frame deletion of cpxR, which encodes the cognate response regulator, displayed an enhanced ability to interact with cell monolayers, as well as elevated inv and rovA transcription. This phenotype could be drastically suppressed by providing a wild-type copy of cpxR in trans. We propose a mechanism of inv regulation influenced by the direct negative effects of phosphorylated CpxR on inv and rovA transcription. In this fashion, sensing of extracytoplasmic stress by CpxAR contributes to productive Yersinia sp.-eukaryotic cell interactions.
Figures




References
-
- Bengoechea, J. A., L. Zhang, P. Toivanen, and M. Skurnik. 2002. Regulatory network of lipopolysaccharide O-antigen biosynthesis in Yersinia enterocolitica includes cell envelope-dependent signals. Mol. Microbiol. 44:1045-1062. - PubMed
-
- Bengoechea, J. A., H. Najdenski, and M. Skurnik. 2004. Lipopolysaccharide O antigen status of Yersinia enterocolitica O:8 is essential for virulence and absence of O antigen affects the expression of other Yersinia virulence factors. Mol. Microbiol. 52:451-469. - PubMed
-
- Butler, S. M., R. A. Festa, M. J. Pearce, and K. H. Darwin. 2006. Self-compartmentalized bacterial proteases and pathogenesis. Mol. Microbiol. 60:553-562. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

