All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation
- PMID: 17620363
- PMCID: PMC2118687
- DOI: 10.1084/jem.20070719
All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation
Abstract
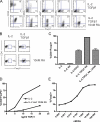
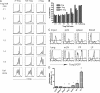
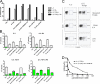
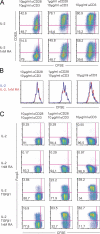
We demonstrate that all-trans retinoic acid (RA) induces FoxP3(+) adaptive T regulatory cells (A-Tregs) to acquire a gut-homing phenotype (alpha 4 beta 7(+) CC chemokine receptor 9(+)) and the capacity to home to the lamina propria of the small intestine. Under conditions that favor the differentiation of A-Tregs (transforming growth factor-beta1 and interleukin 2) in vitro, the inclusion of RA induces nearly all activated CD4(+) T cells to express FoxP3 and greatly increases the accumulation of these cells. In the absence of RA, A-Treg differentiation is abruptly impaired by proficient antigen presenting cells or through direct co-stimulation. In the presence of RA, A-Treg generation occurs even in the presence of high levels of co-stimulation, with RA attenuating co-stimulation from interfering from FoxP3 induction. The recognition that RA induces gut imprinting, together with our finding that it enhances A-Treg conversion, differentiation, and expansion, indicates that RA production in vivo may drive both the imprinting and A-Treg development in the face of overt inflammation.
Figures
Comment in
-
Oral tolerance: is it all retinoic acid?J Exp Med. 2007 Aug 6;204(8):1737-9. doi: 10.1084/jem.20071251. Epub 2007 Jul 9. J Exp Med. 2007. PMID: 17620364 Free PMC article.
References
-
- Izcue, A., J.L. Coombes, and F. Powrie. 2006. Regulatory T cells suppress systemic and mucosal immune activation to control intestinal inflammation. Immunol. Rev. 212:256–271. - PubMed
-
- Bouma, G., and W. Strober. 2003. The immunological and genetic basis of inflammatory bowel disease. Nat. Rev. Immunol. 3:521–533. - PubMed
-
- Mora, J.R., M.R. Bono, N. Manjunath, W. Weninger, L.L. Cavanagh, M. Rosemblatt, and U.H. von Andrian. 2003. Selective imprinting of gut-homing T cells by Peyer's patch dendritic cells. Nature. 424:88–93. - PubMed
-
- Berlin, C., E.L. Berg, M.J. Briskin, D.P. Andrew, P.J. Kilshaw, B. Holzmann, I.L. Weissman, A. Hamann, and E.C. Butcher. 1993. Alpha 4 beta 7 integrin mediates lymphocyte binding to the mucosal vascular addressin MAdCAM-1. Cell. 74:185–195. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

