Human type 2 17 beta-hydroxysteroid dehydrogenase mRNA and protein distribution in placental villi at mid and term pregnancy
- PMID: 17623101
- PMCID: PMC1947994
- DOI: 10.1186/1477-7827-5-30
Human type 2 17 beta-hydroxysteroid dehydrogenase mRNA and protein distribution in placental villi at mid and term pregnancy
Abstract
Background: During human pregnancy, the placental villi produces high amounts of estradiol. This steroid is secreted by the syncytium, which is directly in contact with maternal blood. Estradiol has to cross placental foetal vessels to reach foetal circulation. The enzyme 17beta-hydroxysteroid dehydrogenase type 2 (17beta-HSD2) was detected in placental endothelial cells of foetal vessels inside the villi. This enzyme catalyzes the conversion of estradiol to estrone, and of testosterone to androstenedione. It was proposed that estradiol level into foetal circulation could be regulated by 17beta-HSD2.
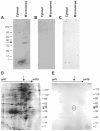
Methods: We obtained placentas from 10 to 26 6/7 weeks of pregnancy from women undergoing voluntary termination of pregnancy, term placentas were collected after normal spontaneous vaginal deliveries. We quantified 17beta-HSD2 mRNA levels in mid-gestation and term human placenta by RT-QPCR. We produced a new anti-17beta-HSD2 antibody to study its spatio-temporal expression by immunohistochemistry. We also compared steroid levels (testosterone, estrone and estradiol) and 17beta-HSD2 mRNA and protein levels between term placenta and endometrium.
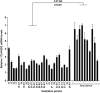
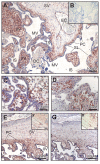
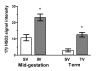
Results: High 17beta-HSD2 mRNA and protein levels were found in both mid-gestation and term placentas. However, we showed that 17beta-HSD2 mRNA levels increase by 2.27 fold between mid-gestation and term. This period coincides with a transitional phase in the development of the villous vasculature. In mid-gestation placenta, high levels of 17beta-HSD2 were found in mesenchymal villi and immature intermediate villi, more precisely in endothelial cells of the stromal channel. At term, high levels of 17beta-HSD2 were found in the numerous sinusoidal capillaries of terminal villi. 17beta-HSD2 mRNA and protein levels in term placentas were respectively 25.4 fold and 30 to 60 fold higher than in the endometrium. Steroid levels were also significantly higher in term placenta than in the endometrium.
Conclusion: The spatial and temporal expression of 17beta-HSD2 in the placenta during pregnancy and the comparison of 17beta-HSD2 expression and steroid levels between placental villi and endometrium are compatible with a role in the modulation of active and inactive forms of estrogens. Our observations strongly support the hypothesis that 17beta-HSD2 acts as a barrier decreasing estradiol secretion rates in the foetal circulation.
Figures


Similar articles
-
Localization of type 1 17beta-hydroxysteroid dehydrogenase mRNA and protein in syncytiotrophoblasts and invasive cytotrophoblasts in the human term villi.J Endocrinol. 2000 May;165(2):217-22. doi: 10.1677/joe.0.1650217. J Endocrinol. 2000. PMID: 10810285
-
Course of placental 11beta-hydroxysteroid dehydrogenase type 2 and 15-hydroxyprostaglandin dehydrogenase mRNA expression during human gestation.Eur J Endocrinol. 2001 Aug;145(2):187-92. doi: 10.1530/eje.0.1450187. Eur J Endocrinol. 2001. PMID: 11454515
-
Expression of 17beta-hydroxysteroid dehydrogenase type 2 in pelvic endometriosis.Gynecol Endocrinol. 2007 Apr;23(4):188-92. doi: 10.1080/09513590701200850. Gynecol Endocrinol. 2007. PMID: 17505937
-
17Beta-hydroxysteroid dehydrogenases in human endometrium and its disorders.Mol Cell Endocrinol. 2006 Mar 27;248(1-2):136-40. doi: 10.1016/j.mce.2005.11.038. Epub 2006 Jan 6. Mol Cell Endocrinol. 2006. PMID: 16406263 Review.
-
Estrogen metabolism and malignancy: analysis of the expression and function of 17beta-hydroxysteroid dehydrogenases in colonic cancer.Mol Cell Endocrinol. 2001 Jan 22;171(1-2):53-60. doi: 10.1016/s0303-7207(00)00418-4. Mol Cell Endocrinol. 2001. PMID: 11165011 Review.
Cited by
-
Advanced multi-modal mass spectrometry imaging reveals functional differences of placental villous compartments at microscale resolution.Nat Commun. 2025 Feb 28;16(1):2061. doi: 10.1038/s41467-025-57107-y. Nat Commun. 2025. PMID: 40021619 Free PMC article.
-
Effect of tumor necrosis factor-alpha on estrogen metabolism and endometrial cells: potential physiological and pathological relevance.J Clin Endocrinol Metab. 2009 Jan;94(1):285-93. doi: 10.1210/jc.2008-1389. Epub 2008 Oct 28. J Clin Endocrinol Metab. 2009. PMID: 18957495 Free PMC article.
-
Physiology and Pathophysiology of Steroid Biosynthesis, Transport and Metabolism in the Human Placenta.Front Pharmacol. 2018 Sep 12;9:1027. doi: 10.3389/fphar.2018.01027. eCollection 2018. Front Pharmacol. 2018. PMID: 30258364 Free PMC article. Review.
-
Steroidogenic enzyme and key decidualization marker dysregulation in endometrial stromal cells from women with versus without endometriosis.Biol Reprod. 2009 Jan;80(1):105-14. doi: 10.1095/biolreprod.108.070300. Epub 2008 Sep 24. Biol Reprod. 2009. PMID: 18815356 Free PMC article.
-
Levels of dihydrotestosterone, testosterone, androstenedione, and estradiol in canalicular, saccular, and alveolar mouse lungs.Lung. 2010 Jun;188(3):229-33. doi: 10.1007/s00408-010-9231-x. Epub 2010 Feb 17. Lung. 2010. PMID: 20162289
References
-
- Albrecht ED, Pepe GJ. Placental steroid hormone biosynthesis in primate pregnancy. Endocr Rev. 1990;11(1):124–150. - PubMed
-
- Khosla SS, Rooney SA. Stimulation of fetal lung surfactant production by administration of estradiol to the maternal rabbit. Am J Obstet Gynecol. 1979;133:213–216. - PubMed
-
- Pepe GJ, Waddell BJ, Albrecht ED. Activation of the baboon fetal hypothalamic-pituitary-adrenocortical axis at midgestation by estrogen-induced changes in placental corticosteroid metabolism. Endocrinology. 1990;127(6):3117–3123. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

