Angiogenesis is not impaired in connective tissue growth factor (CTGF) knock-out mice
- PMID: 17625227
- PMCID: PMC3957533
- DOI: 10.1369/jhc.7A7258.2007
Angiogenesis is not impaired in connective tissue growth factor (CTGF) knock-out mice
Abstract
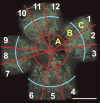
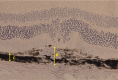
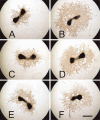
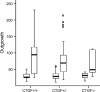
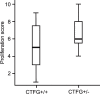
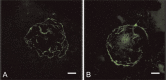
Connective tissue growth factor (CTGF) is a member of the CCN family of growth factors. CTGF is important in scarring, wound healing, and fibrosis. It has also been implicated to play a role in angiogenesis, in addition to vascular endothelial growth factor (VEGF). In the eye, angiogenesis and subsequent fibrosis are the main causes of blindness in conditions such as diabetic retinopathy. We have applied three different models of angiogenesis to homozygous CTGF(-/-) and heterozygous CTGF(+/-) mice to establish involvement of CTGF in neovascularization. CTGF(-/-) mice die around birth. Therefore, embryonic CTGF(-/-), CTGF(+/-), and CTGF(+/+) bone explants were used to study in vitro angiogenesis, and neonatal and mature CTGF(+/-) and CTGF(+/+) mice were used in models of oxygen-induced retinopathy and laser-induced choroidal neovascularization. Angiogenesis in vitro was independent of the CTGF genotype in both the presence and the absence of VEGF. Oxygen-induced vascular pathology in the retina, as determined semi-quantitatively, and laser-induced choroidal neovascularization, as determined quantitatively, were also not affected by the CTGF genotype. Our data show that downregulation of CTGF levels does not affect neovascularization, indicating distinct roles of VEGF and CTGF in angiogenesis and fibrosis in eye conditions.
Figures


Similar articles
-
Regulation of angiogenesis and endothelial cell function by connective tissue growth factor (CTGF) and cysteine-rich 61 (CYR61).Angiogenesis. 2002;5(3):153-65. doi: 10.1023/a:1023823803510. Angiogenesis. 2002. PMID: 12831056
-
A role for connective tissue growth factor in the pathogenesis of choroidal neovascularization.Arch Ophthalmol. 2003 Sep;121(9):1283-8. doi: 10.1001/archopht.121.9.1283. Arch Ophthalmol. 2003. PMID: 12963611
-
The angio-fibrotic switch of VEGF and CTGF in proliferative diabetic retinopathy.PLoS One. 2008 Jul 16;3(7):e2675. doi: 10.1371/journal.pone.0002675. PLoS One. 2008. PMID: 18628999 Free PMC article.
-
Role of the vascular endothelial growth factor isoforms in retinal angiogenesis and DiGeorge syndrome.Verh K Acad Geneeskd Belg. 2005;67(4):229-76. Verh K Acad Geneeskd Belg. 2005. PMID: 16334858 Review.
-
CTGF/Hcs24 as a multifunctional growth factor for fibroblasts, chondrocytes and vascular endothelial cells.Drug News Perspect. 2003 Jan-Feb;16(1):11-21. doi: 10.1358/dnp.2003.16.1.829302. Drug News Perspect. 2003. PMID: 12682668 Review.
Cited by
-
CTGF disrupts alveolarization and induces pulmonary hypertension in neonatal mice: implication in the pathogenesis of severe bronchopulmonary dysplasia.Am J Physiol Lung Cell Mol Physiol. 2011 Mar;300(3):L330-40. doi: 10.1152/ajplung.00270.2010. Epub 2011 Jan 14. Am J Physiol Lung Cell Mol Physiol. 2011. PMID: 21239535 Free PMC article.
-
Transplanted Umbilical Cord Mesenchymal Stem Cells Modify the In Vivo Microenvironment Enhancing Angiogenesis and Leading to Bone Regeneration.Stem Cells Dev. 2015 Jul 1;24(13):1570-81. doi: 10.1089/scd.2014.0490. Epub 2015 Mar 18. Stem Cells Dev. 2015. PMID: 25685989 Free PMC article.
-
Increased expression of CCN2 in the red flashing light-induced myopia in guinea pigs.Biomed Res Int. 2013;2013:761823. doi: 10.1155/2013/761823. Epub 2013 Jul 14. Biomed Res Int. 2013. PMID: 23936844 Free PMC article.
-
Functional role of periostin in development and wound repair: implications for connective tissue disease.J Cell Commun Signal. 2008 Jun;2(1-2):9-17. doi: 10.1007/s12079-008-0023-5. Epub 2008 Jul 20. J Cell Commun Signal. 2008. PMID: 18642132 Free PMC article.
-
CYR61/CCN1 Regulates Sclerostin Levels and Bone Maintenance.J Bone Miner Res. 2018 Jun;33(6):1076-1089. doi: 10.1002/jbmr.3394. Epub 2018 Mar 5. J Bone Miner Res. 2018. PMID: 29351359 Free PMC article.
References
-
- Agostini H, Boden K, Unsold A, Martin G, Hansen L, Fiedler U, Esser N, et al. (2005) A single local injection of recombinant VEGF receptor 2 but not of Tie2 inhibits retinal neovascularization in the mouse. Curr Eye Res 30: 249–257 - PubMed
-
- Aiello LP, Gardner TW, King GL, Blankenship G, Cavallerano JD, Ferris FL, Klein R. (1998) Diabetic retinopathy. Diabetes Care 21: 143–156 - PubMed
-
- Campochiaro PA, Hackett SF. (2003) Ocular neovascularization: a valuable model system. Oncogene 22: 6537–6548 - PubMed
-
- Chang CC, Lin MT, Lin BR, Jeng YM, Chen ST, Chu CY, Chen RJ, et al. (2006) Effect of connective tissue growth factor on hypoxia-inducible factor 1α degradation and tumor angiogenesis. J Natl Cancer Inst 98: 984–995 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

