Alcohol inhibits NR2B-containing NMDA receptors in the ventral bed nucleus of the stria terminalis
- PMID: 17625498
- PMCID: PMC2864636
- DOI: 10.1038/sj.npp.1301504
Alcohol inhibits NR2B-containing NMDA receptors in the ventral bed nucleus of the stria terminalis
Abstract
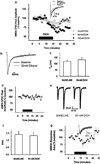
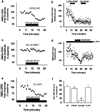
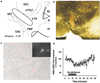
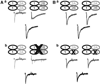
Components of the mesolimbic dopamine system, in particular dopaminergic cells in the ventral tegmental area (VTA), have been implicated in the acute reinforcing actions of ethanol. The ventral bed nucleus of the stria terminalis (vBNST) potently regulates dopaminergic cell firing in the VTA, and has been implicated in the behavioral actions of ethanol. The N-methyl-D-asparate receptor (NMDAR) is a major molecular target of ethanol, however, current evidence suggests that ethanol regulation of NMDAR function is widely variable and likely depends on a number of factors. Thus, it is critical to investigate ethanol regulation of NMDAR function at synapses relevant to ethanol-regulated behaviors, such as in the vBNST. Here we show, using multiple techniques, that ethanol inhibits NMDAR function in vBNST neurons in a postsynaptic fashion. Further, we demonstrate the functional presence of both NR2A and NR2B-containing NMDARs in the vBNST. While genetic removal of NR2A did not alter the magnitude of ethanol inhibition, pharmacological blockade of NR2B rendered synaptically activated NMDARs insensitive to ethanol inhibition. Finally, we demonstrate that ethanol inhibits NMDARs in cells in the vBNST that project to the VTA, providing a direct means by which ethanol in the vBNST can modulate the dopaminergic system.
Conflict of interest statement
Dr Kash declares that, except for income received from primary employer, no financial support or compensation has been received from any individual or corporate entity over the past 3 years for research or professional service. Dr Matthews received compensation from Meharry Medical College. Dr Winder received a distribution from Columbia University for the licensing of transgenic mouse technology to Memory Pharmaceuticals and received a consultancy fee from MEDAcorp. All of the authors declare that there are no personal financial holdings that could be perceived as constituting a potential conflict of interest.
Figures


Similar articles
-
Ethanol effects on N-methyl-D-aspartate receptors in the bed nucleus of the stria terminalis.Cold Spring Harb Perspect Med. 2013 Apr 1;3(4):a012161. doi: 10.1101/cshperspect.a012161. Cold Spring Harb Perspect Med. 2013. PMID: 23426579 Free PMC article. Review.
-
Alcohol exposure alters NMDAR function in the bed nucleus of the stria terminalis.Neuropsychopharmacology. 2009 Oct;34(11):2420-9. doi: 10.1038/npp.2009.69. Epub 2009 Jun 24. Neuropsychopharmacology. 2009. PMID: 19553918 Free PMC article.
-
Activation of ventral tegmental area cells by the bed nucleus of the stria terminalis: a novel excitatory amino acid input to midbrain dopamine neurons.J Neurosci. 2002 Jun 15;22(12):5173-87. doi: 10.1523/JNEUROSCI.22-12-05173.2002. J Neurosci. 2002. PMID: 12077212 Free PMC article.
-
Dual synaptic sites of D(1)-dopaminergic regulation of ethanol sensitivity of NMDA receptors in nucleus accumbens.Synapse. 2005 Oct;58(1):30-44. doi: 10.1002/syn.20181. Synapse. 2005. PMID: 16037948
-
Ethanol-mediated facilitation of AMPA receptor function in the dorsomedial striatum: implications for alcohol drinking behavior.J Neurosci. 2012 Oct 24;32(43):15124-32. doi: 10.1523/JNEUROSCI.2783-12.2012. J Neurosci. 2012. PMID: 23100433 Free PMC article.
Cited by
-
A corticotropin releasing factor pathway for ethanol regulation of the ventral tegmental area in the bed nucleus of the stria terminalis.J Neurosci. 2013 Jan 16;33(3):950-60. doi: 10.1523/JNEUROSCI.2949-12.2013. J Neurosci. 2013. PMID: 23325234 Free PMC article.
-
Ketamine administration during a critical period after forced ethanol abstinence inhibits the development of time-dependent affective disturbances.Neuropsychopharmacology. 2018 Aug;43(9):1915-1923. doi: 10.1038/s41386-018-0102-0. Epub 2018 May 24. Neuropsychopharmacology. 2018. PMID: 29907878 Free PMC article.
-
Synaptic targets: Chronic alcohol actions.Neuropharmacology. 2017 Aug 1;122:85-99. doi: 10.1016/j.neuropharm.2017.01.013. Epub 2017 Jan 17. Neuropharmacology. 2017. PMID: 28108359 Free PMC article. Review.
-
Glutamatergic targets for new alcohol medications.Psychopharmacology (Berl). 2013 Oct;229(3):539-54. doi: 10.1007/s00213-013-3226-2. Epub 2013 Sep 1. Psychopharmacology (Berl). 2013. PMID: 23995381 Free PMC article. Review.
-
Ethanol effects on N-methyl-D-aspartate receptors in the bed nucleus of the stria terminalis.Cold Spring Harb Perspect Med. 2013 Apr 1;3(4):a012161. doi: 10.1101/cshperspect.a012161. Cold Spring Harb Perspect Med. 2013. PMID: 23426579 Free PMC article. Review.
References
-
- Bartlett TE, Bannister NJ, Collett VJ, Dargan SL, Massey PV, Bortolotto ZA, et al. Differential roles of NR2A and NR2B-containing NMDA receptors in LTP and LTD in the CA1 region of two-week old rat hippocampus. Neuropharmacology. 2006;52:60–70. - PubMed
-
- Boyce-Rustay JM, Holmes A. Functional roles of NMDA receptor NR2A and NR2B subunits in the acute intoxicating effects of ethanol in mice. Synapse. 2005;56:222–225. - PubMed
-
- Brodie MS, Pesold C, Appel SB. Ethanol directly excites dopaminergic ventral tegmental area reward neurons. Alcohol Clin Exp Res. 1999;23:1848–1852. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

