Modulation of heme redox potential in the cytochrome c6 family
- PMID: 17625855
- PMCID: PMC7610927
- DOI: 10.1021/ja072346g
Modulation of heme redox potential in the cytochrome c6 family
Abstract
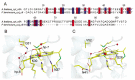
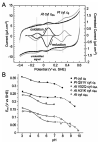
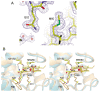
Cytochrome c6A is a unique dithio-cytochrome of green algae and plants. It has a very similar core structure to that of bacterial and algal cytochromes c6 but is unable to fulfill the same function of transferring electrons from cytochrome f to photosystem I. A key feature is that its heme midpoint potential is more than 200 mV below that of cytochrome c6 despite having His and Met as axial heme-iron ligands. To identify the molecular origins of the difference in potential, the structure of cytochrome c6 from the cyanobacterium Phormidium laminosum has been determined by X-ray crystallography and compared with the known structure of cytochrome c6A. One salient difference of the heme pockets is that a highly conserved Gln (Q51) in cytochrome c6 is replaced by Val (V52) in c6A. Using protein film voltammetry, we found that swapping these residues raised the c6A potential by +109 mV and decreased that of c6 by almost the same extent, -100 mV. X-ray crystallography of the V52Q protein showed that the Gln residue adopts the same configuration relative to the heme as in cytochrome c6 and we propose that this stereochemistry destabilizes the oxidized form of the heme. Consequently, replacement of Gln by Val was probably a key step in the evolution of cytochrome c6A from cytochrome c6, inhibiting reduction by the cytochrome b6f complex and facilitating establishment of a new function.
Figures
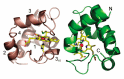
Similar articles
-
Cytochrome c6A: discovery, structure and properties responsible for its low haem redox potential.Biochem Soc Trans. 2008 Dec;36(Pt 6):1175-9. doi: 10.1042/BST0361175. Biochem Soc Trans. 2008. PMID: 19021519 Review.
-
Structure of cytochrome c6A, a novel dithio-cytochrome of Arabidopsis thaliana, and its reactivity with plastocyanin: implications for function.J Mol Biol. 2006 Jul 28;360(5):968-77. doi: 10.1016/j.jmb.2006.05.065. Epub 2006 Jun 16. J Mol Biol. 2006. PMID: 16815443
-
Structural and kinetic studies of imidazole binding to two members of the cytochrome c (6) family reveal an important role for a conserved heme pocket residue.J Biol Inorg Chem. 2011 Apr;16(4):577-88. doi: 10.1007/s00775-011-0758-y. Epub 2011 Jan 26. J Biol Inorg Chem. 2011. PMID: 21267610
-
Cytochrome c(6B) of Synechococcus sp. WH 8102--crystal structure and basic properties of novel c(6)-like family representative.Biochem Biophys Res Commun. 2014 Jan 24;443(4):1131-5. doi: 10.1016/j.bbrc.2013.10.167. Epub 2013 Nov 9. Biochem Biophys Res Commun. 2014. PMID: 24216109
-
The novel cytochrome c6 of chloroplasts: a case of evolutionary bricolage?J Exp Bot. 2006;57(1):13-22. doi: 10.1093/jxb/erj023. Epub 2005 Nov 29. J Exp Bot. 2006. PMID: 16317035 Review.
Cited by
-
Purification and characterization of cytochrome c(6) from Acaryochloris marina.Photosynth Res. 2009 Oct;102(1):43-51. doi: 10.1007/s11120-009-9482-7. Epub 2009 Aug 13. Photosynth Res. 2009. PMID: 19680778 Free PMC article.
-
Structural basis for the oxidation of protein-bound sulfur by the sulfur cycle molybdohemo-enzyme sulfane dehydrogenase SoxCD.J Biol Chem. 2011 Mar 11;286(10):8349-8360. doi: 10.1074/jbc.M110.193631. Epub 2010 Dec 8. J Biol Chem. 2011. PMID: 21147779 Free PMC article.
-
Metalloproteins containing cytochrome, iron-sulfur, or copper redox centers.Chem Rev. 2014 Apr 23;114(8):4366-469. doi: 10.1021/cr400479b. Chem Rev. 2014. PMID: 24758379 Free PMC article. Review. No abstract available.
-
Soluble domains of cytochrome c-556 and Rieske iron-sulfur protein from Chlorobaculum tepidum: Crystal structures and interaction analysis.Curr Res Struct Biol. 2023 Apr 19;5:100101. doi: 10.1016/j.crstbi.2023.100101. eCollection 2023. Curr Res Struct Biol. 2023. PMID: 37180033 Free PMC article.
-
Comparison of the backbone dynamics of wild-type Hydrogenobacter thermophilus cytochrome c(552) and its b-type variant.J Biomol NMR. 2015 Jun;62(2):221-31. doi: 10.1007/s10858-015-9938-3. Epub 2015 May 8. J Biomol NMR. 2015. PMID: 25953310 Free PMC article.
References
-
- Tezcan FA, Winkler JR, Gray HB. J Am Chem Soc. 1998;120:13383–13388.
-
- Berghuis AM, Brayer GD. J Mol Biol. 1992;223:959–976. - PubMed
-
- Berghuis AM, Guillemette JG, McLendon G, Sherman F, Smith M, Brayer GD. J Mol Biol. 1994;236:786–799. - PubMed
-
- Schnackenberg J, Than ME, Mann K, Wiegand G, Huber R, Reuter W. J Mol Biol. 1999;290:1019–1030. - PubMed
-
- Marcaida MJ, Schlarb-Ridley BG, Worrall JAR, Wastl J, Evans TJ, Bendall DS, Luisi BF, Howe CJ. J Mol Biol. 2006;360:968–977. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

