Lesions of the tegmentomammillary circuit in the head direction system disrupt the head direction signal in the anterior thalamus
- PMID: 17626218
- PMCID: PMC6672597
- DOI: 10.1523/JNEUROSCI.0268-07.2007
Lesions of the tegmentomammillary circuit in the head direction system disrupt the head direction signal in the anterior thalamus
Abstract
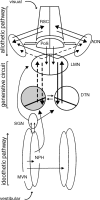
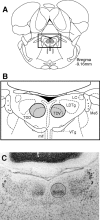
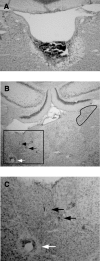
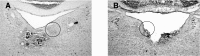
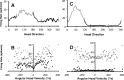
Head direction (HD) cells in the rodent limbic system are believed to correspond to a cognitive representation of directional heading in the environment. Lesions of vestibular hair cells disrupt the characteristic firing patterns of HD cells, and thus vestibular afference is a critical contributor to the HD signal. A subcortical pathway that may convey this information includes the dorsal tegmental nucleus of Gudden (DTN) and the lateral mammillary nucleus (LMN). To test the hypothesis that the DTN and LMN are critical components for generating HD cell activity, we made electrolytic lesions of the DTN or LMN in rats and screened for HD cell activity in the anterior thalamus. Directional activity was absent in all animals with complete LMN lesions and in animals with complete DTN lesions, although a few HD cells were isolated in animals with incomplete lesions. Some DTN-lesioned animals contained cells whose firing rates were modulated by angular head velocity. Although cells with bursting patterns of activity have been observed in the anterior dorsal nucleus of the thalamus of animals with disruption of vestibular inputs, this pattern of activity was not observed in either the LMN- or DTN-lesioned animals. The general absence of direction-specific activity in the anterior thalamus of animals with DTN or LMN lesions is consistent with the view that the DTN-LMN circuit is essential for the generation of HD cell activity.
Figures

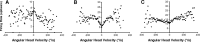


References
-
- Allen GV, Hopkins DA. Mamillary body in the rat: topography and synaptology of projections from the subicular complex, prefrontal cortex, and midbrain tegmentum. J Comp Neurol. 1989;286:311–336. - PubMed
-
- Allen GV, Hopkins DA. Topography and synaptology of mamillary body projections to the mesencephalon and pons in the rat. J Comp Neurol. 1990;301:214–231. - PubMed
-
- Bassett JP, Taube JS. Head direction signal generation: ascending and descending information streams. In: Wiener SI, Taube JS, editors. Head direction cells and the neural mechanisms of spatial orientation. Cambridge, MA: MIT; 2005. pp. 83–109.
-
- Batschelet E. New York: Academic; 1981. Circular statistics in biology.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
