Low-frequency stimulation induces a pathway-specific late phase of LTP in the amygdala that is mediated by PKA and dependent on protein synthesis
- PMID: 17626908
- PMCID: PMC1934345
- DOI: 10.1101/lm.593407
Low-frequency stimulation induces a pathway-specific late phase of LTP in the amygdala that is mediated by PKA and dependent on protein synthesis
Abstract
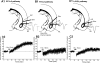
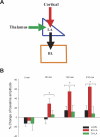
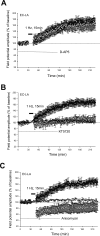
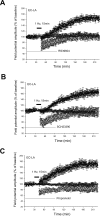
Activity-dependent changes in synaptic efficacy are thought to be the key cellular mechanism for the formation and storage of both explicit and implicit memory. Different patterns of stimulation can elicit different changes in the efficiency on excitatory synaptic transmission. Here, we examined the synaptic changes in the amygdala of adult mice produced by low-frequency stimulation (1 Hz, 15 min, LFS). We first compared the synaptic changes induced by LFS in three different synaptic pathways of amygdala: cortical-lateral amygdala, thalamic-lateral amygdala, and lateral-basolateral amygdala pathways. We find that the plastic changes induced by LFS are different between synaptic pathways. Low-frequency stimulation selectively elicits a slow onset and protein synthesis-dependent late-phase LTP in the cortical-lateral amygdala pathway, but not in the thalamic-lateral or lateral-basolateral pathways. We next analyzed LTP induced by LFS in the cortical-lateral amygdala pathway and found that three PKA-coupling neurotransmitter receptors are involved: 5-HT4, Dopamine D1, and beta-adrenergic receptors. Antagonists of these receptors block the LFS L-LTP, but the effects of agonists of these receptors are clearly different. These results indicate that the threshold for the induction of LFS L-LTP is different among these pathways and that the maintenance of LFS L-LTP requires a cross-talk among multiple neurotransmitters.
Figures
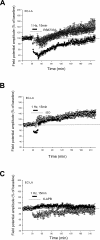
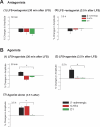
Similar articles
-
Stabilization of thalamo-cortical long-term potentiation by the amygdala: cholinergic and transcription-dependent mechanisms.Eur J Neurosci. 2004 Jul;20(2):557-65. doi: 10.1111/j.1460-9568.2004.03515.x. Eur J Neurosci. 2004. PMID: 15233765
-
Both protein kinase A and mitogen-activated protein kinase are required in the amygdala for the macromolecular synthesis-dependent late phase of long-term potentiation.J Neurosci. 2000 Sep 1;20(17):6317-25. doi: 10.1523/JNEUROSCI.20-17-06317.2000. J Neurosci. 2000. PMID: 10964936 Free PMC article.
-
Low-frequency stimulation induces a new form of LTP, metabotropic glutamate (mGlu5) receptor- and PKA-dependent, in the CA1 area of the rat hippocampus.Hippocampus. 2006;16(4):345-60. doi: 10.1002/hipo.20146. Hippocampus. 2006. PMID: 16302229
-
Cooperation of taurine uptake and dopamine D1 receptor activation facilitates the induction of protein synthesis-dependent late LTP.Neuropharmacology. 2014 Apr;79:101-11. doi: 10.1016/j.neuropharm.2013.10.035. Epub 2013 Nov 10. Neuropharmacology. 2014. PMID: 24225198
-
Perirhinal cortex and thalamic stimulation induces LTP in different areas of the amygdala.Ann N Y Acad Sci. 2000 Jun;911:474-6. doi: 10.1111/j.1749-6632.2000.tb06749.x. Ann N Y Acad Sci. 2000. PMID: 10911897 Review. No abstract available.
Cited by
-
Noradrenergic innervation of pyramidal cells in the rat basolateral amygdala.Neuroscience. 2013 Jan 3;228:395-408. doi: 10.1016/j.neuroscience.2012.10.035. Epub 2012 Oct 26. Neuroscience. 2013. PMID: 23103792 Free PMC article.
-
Fear learning and extinction are linked to neuronal plasticity through Rin1 signaling.J Neurosci Res. 2010 Mar;88(4):917-26. doi: 10.1002/jnr.22252. J Neurosci Res. 2010. PMID: 19830836 Free PMC article.
-
Modification of Fear Memory by Pharmacological and Behavioural Interventions during Reconsolidation.PLoS One. 2016 Aug 18;11(8):e0161044. doi: 10.1371/journal.pone.0161044. eCollection 2016. PLoS One. 2016. PMID: 27537364 Free PMC article. Clinical Trial.
-
Long-Term Potentiation at CA3-CA1 Hippocampal Synapses with Special Emphasis on Aging, Disease, and Stress.Front Aging Neurosci. 2011 May 20;3:7. doi: 10.3389/fnagi.2011.00007. eCollection 2011. Front Aging Neurosci. 2011. PMID: 21647396 Free PMC article.
-
PKA has a critical role in synaptic delivery of GluR1- and GluR4-containing AMPARs during initial stages of acquisition of in vitro classical conditioning.J Neurophysiol. 2009 May;101(5):2539-49. doi: 10.1152/jn.91282.2008. Epub 2009 Mar 4. J Neurophysiol. 2009. PMID: 19261706 Free PMC article.
References
-
- Amorapanth P., Ledoux J.E., Nader K. Different lateral amygdala outputs mediate reactions and actions elicited by a fear-arousing stimulus. Nat. Neurosci. 2000;3:74–79. - PubMed
-
- Asan E. The catecholaminergic innervation of the rat amygdala. Adv. Anat. Embryo. Cell Biol. 1998;142:1–118. - PubMed
-
- Bach M.E., Barad M., Son H., Zhuo M., Lu Y.-F., Shih R., Mansuy I., Hawkins R.D., Kandel E.R. Age-related defects in spatial memory are correlated with defects in the late phase of hippocampal long-term potentiation in vitro and attenuated by drugs that enhance the cAMP signaling pathway. Proc. Natl. Acad. Sci. 1999;96:5280–5285. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
