Extracellular ATP is cytotoxic to mononuclear phagocytes but does not induce killing of intracellular Mycobacterium avium subsp. paratuberculosis
- PMID: 17634511
- PMCID: PMC2043304
- DOI: 10.1128/CVI.00166-07
Extracellular ATP is cytotoxic to mononuclear phagocytes but does not induce killing of intracellular Mycobacterium avium subsp. paratuberculosis
Abstract
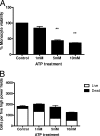
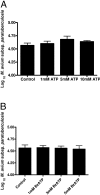
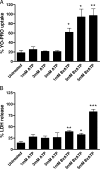
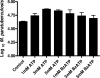
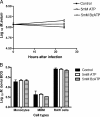
Mycobacterium avium subsp. paratuberculosis is the etiologic agent of Johne's disease, a chronic granulomatous enteritis in ruminants. ATP has been reported to induce cell death of macrophages and killing of Mycobacterium species in human and murine macrophages. In this study we investigated the short-term effect of ATP on the viability of M. avium subsp. paratuberculosis-infected bovine mononuclear phagocytes and the bacilli within them. Addition of 5 mM ATP to M. avium subsp. paratuberculosis-infected bovine monocytes resulted in 50% cytotoxicity of bovine monocytes at 24 h. Addition of 2'(3')-O-(4-benzoylbenzoyl) ATP triethylammonium salt (Bz-ATP), which is a longer-lived ATP homologue and purinergic receptor agonist, significantly increased the uptake of YO-PRO, which is a marker for membrane pore activation by P2X receptors. Addition of Bz-ATP also stimulated lactate dehydrogenase release and caspase-3 activity in infected bovine monocytes. Neither ATP nor Bz-ATP reduced the survival of M. avium subsp. paratuberculosis in bovine mononuclear phagocytes. Likewise, addition of ATP or Bz-ATP was cytotoxic to murine macrophage cell lines (RAW 264.7 and J774A.1 cells) but did not affect the intracellular survival of M. avium subsp. paratuberculosis, nor were the numbers of viable Mycobacterium avium subsp. avium or Mycobacterium bovis BCG cells altered in bovine mononuclear phagocytes or J774A.1 cells following ATP or Bz-ATP treatment. These data suggest that extracellular ATP does not induce the killing of intracellular M. avium subsp. paratuberculosis in bovine mononuclear phagocytes.
Figures
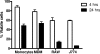
Similar articles
-
Tactics of Mycobacterium avium subsp. paratuberculosis for intracellular survival in mononuclear phagocytes.J Vet Sci. 2008 Mar;9(1):1-8. doi: 10.4142/jvs.2008.9.1.1. J Vet Sci. 2008. PMID: 18296882 Free PMC article. Review.
-
ATP release by infected bovine monocytes increases the intracellular survival of Mycobacterium avium subsp. paratuberculosis.Comp Immunol Microbiol Infect Dis. 2009 Sep;32(5):365-77. doi: 10.1016/j.cimid.2007.11.003. Epub 2008 Feb 19. Comp Immunol Microbiol Infect Dis. 2009. PMID: 18243311
-
Use of Hoechst 33342 staining to detect apoptotic changes in bovine mononuclear phagocytes infected with Mycobacterium avium subsp. paratuberculosis.Clin Diagn Lab Immunol. 2001 Mar;8(2):460-4. doi: 10.1128/CDLI.8.2.460-464.2001. Clin Diagn Lab Immunol. 2001. PMID: 11238240 Free PMC article.
-
Bovine monocyte TLR2 receptors differentially regulate the intracellular fate of Mycobacterium avium subsp. paratuberculosis and Mycobacterium avium subsp. avium.J Leukoc Biol. 2008 Jan;83(1):48-55. doi: 10.1189/jlb.0707490. Epub 2007 Oct 3. J Leukoc Biol. 2008. PMID: 17913973
-
Review paper: modulation of mononuclear phagocyte function by Mycobacterium avium subsp. paratuberculosis.Vet Pathol. 2008 Nov;45(6):829-41. doi: 10.1354/vp.45-6-829. Vet Pathol. 2008. PMID: 18984786 Review.
Cited by
-
Possible effects of microbial ecto-nucleoside triphosphate diphosphohydrolases on host-pathogen interactions.Microbiol Mol Biol Rev. 2008 Dec;72(4):765-81, Table of Contents. doi: 10.1128/MMBR.00013-08. Microbiol Mol Biol Rev. 2008. PMID: 19052327 Free PMC article. Review.
-
Tactics of Mycobacterium avium subsp. paratuberculosis for intracellular survival in mononuclear phagocytes.J Vet Sci. 2008 Mar;9(1):1-8. doi: 10.4142/jvs.2008.9.1.1. J Vet Sci. 2008. PMID: 18296882 Free PMC article. Review.
-
Extracellular ATP acts on P2Y2 purinergic receptors to facilitate HIV-1 infection.J Exp Med. 2011 Aug 29;208(9):1823-34. doi: 10.1084/jem.20101805. Epub 2011 Aug 22. J Exp Med. 2011. PMID: 21859844 Free PMC article.
References
-
- Aga, M., C. J. Johnson, A. P. Hart, A. G. Guadarrama, M. Suresh, J. Svaren, P. J. Bertics, and B. J. Darien. 2002. Modulation of monocyte signaling and pore formation in response to agonists of the nucleotide receptor P2X(7). J. Leukoc. Biol. 72222-232. - PubMed
-
- Apasov, S. G., M. Koshiba, T. M. Chused, and M. V. Sitkovsky. 1997. Effects of extracellular ATP and adenosine on different thymocyte subsets: possible role of ATP-gated channels and G protein-coupled purinergic receptor. J. Immunol. 1585095-5105. - PubMed
-
- Buell, G., A. D. Michel, C. Lewis, G. Collo, P. P. Humphrey, and A. Surprenant. 1996. P2X1 receptor activation in HL60 cells. Blood 872659-2664. - PubMed
-
- Chacon, O., L. E. Bermudez, and R. G. Barletta. 2004. Johne's disease, inflammatory bowel disease, and Mycobacterium paratuberculosis. Annu. Rev. Microbiol. 58329-363. - PubMed
-
- Chiozzi, P., M. Murgia, S. Falzoni, D. Ferrari, and F. Di Virgilio. 1996. Role of the purinergic P2Z receptor in spontaneous cell death in J774 macrophage cultures. Biochem. Biophys. Res. Commun. 218176-181. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

