Type I gamma phosphatidylinositol phosphate kinase is required for EGF-stimulated directional cell migration
- PMID: 17635937
- PMCID: PMC2064448
- DOI: 10.1083/jcb.200701078
Type I gamma phosphatidylinositol phosphate kinase is required for EGF-stimulated directional cell migration
Abstract
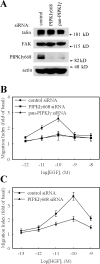
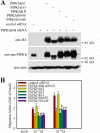
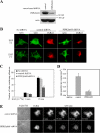
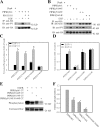
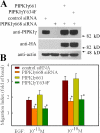
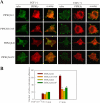
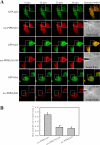
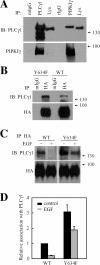
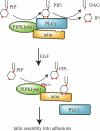
Phosphatidylinositol 4,5-bisphosphate (PI4,5P(2)) modulates a plethora of cytoskeletal interactions that control the dynamics of actin assembly and, ultimately, cell migration. We show that the type Igamma phosphatidylinositol phosphate kinase 661 (PIPKIgamma661), an enzyme that generates PI4,5P(2), is required for growth factor but not G protein-coupled receptor-stimulated directional migration. By generating PI4,5P(2) and regulating talin assembly, PIPKIgamma661 modulates nascent adhesion formation at the leading edge to facilitate cell migration. The epidermal growth factor (EGF) receptor directly phosphorylates PIPKIgamma661 at tyrosine 634, and this event is required for EGF-induced migration. This phosphorylation regulates the interaction between PIPKIgamma661 and phospholipase Cgamma1 (PLCgamma1, an enzyme previously shown to be involved in the regulation of EGF-stimulated migration). Our results suggest that phosphorylation events regulating specific PIPKIgamma661 interactions are required for growth factor-induced migration. These interactions in turn define the spatial and temporal generation of PI4,5P(2) and derived messengers required for directional migration.
Figures

References
-
- Anderson, R.A., and V.T. Marchesi. 1985. Regulation of the association of membrane skeletal protein 4.1 with glycophorin by a polyphosphoinositide. Nature. 318:295–298. - PubMed
-
- Anderson, R.A., I.V. Boronenkov, S.D. Doughman, J. Kunz, and J.C. Loijens. 1999. Phosphatidylinositol phosphate kinases, a multifaceted family of signaling enzymes. J. Biol. Chem. 274:9907–9910. - PubMed
-
- Bairstow, S.F., K. Ling, X. Su, A.J. Firestone, C. Carbonara, and R.A. Anderson. 2006. Type Igamma661 phosphatidylinositol phosphate kinase directly interacts with AP2 and regulates endocytosis. J. Biol. Chem. 281:20632–20642. - PubMed
-
- Borm, B., R.P. Requardt, V. Herzog, and G. Kirfel. 2005. Membrane ruffles in cell migration: indicators of inefficient lamellipodia adhesion and compartments of actin filament reorganization. Exp. Cell Res. 302:83–95. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

