HIV protease inhibitors block the zinc metalloproteinase ZMPSTE24 and lead to an accumulation of prelamin A in cells
- PMID: 17652517
- PMCID: PMC1948915
- DOI: 10.1073/pnas.0704212104
HIV protease inhibitors block the zinc metalloproteinase ZMPSTE24 and lead to an accumulation of prelamin A in cells
Abstract
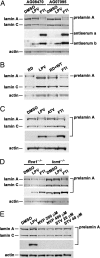
HIV protease inhibitors (HIV-PIs) target the HIV aspartyl protease, which cleaves the HIV gag-pol polyprotein into shorter proteins required for the production of new virions. HIV-PIs are a cornerstone of treatment for HIV but have been associated with lipodystrophy and other side effects. In both human and mouse fibroblasts, we show that HIV-PIs caused an accumulation of prelamin A. The prelamin A in HIV-PI-treated fibroblasts migrated more rapidly than nonfarnesylated prelamin A, comigrating with the farnesylated form of prelamin A that accumulates in ZMPSTE24-deficient fibroblasts. The accumulation of farnesyl-prelamin A in response to HIV-PI treatment was exaggerated in fibroblasts heterozygous for Zmpste24 deficiency. HIV-PIs inhibited the endoproteolytic processing of a GFP-prelamin A fusion protein. The HIV-PIs did not affect the farnesylation of HDJ-2, nor did they inhibit protein farnesyltransferase in vitro. HIV-PIs also did not inhibit the activities of the isoprenyl-cysteine carboxyl methyltransferase ICMT or the prenylprotein endoprotease RCE1 in vitro, but they did inhibit ZMPSTE24 (IC(50): lopinavir, 18.4 +/- 4.6 microM; tipranavir, 1.2 +/- 0.4 microM). We conclude that the HIV-PIs inhibit ZMPSTE24, leading to an accumulation of farnesyl-prelamin A. The inhibition of ZMPSTE24 by HIV-PIs could play a role in the side effects of these drugs.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Comment in
-
HIV protease inhibitors and nuclear lamin processing: getting the right bells and whistles.Proc Natl Acad Sci U S A. 2007 Aug 28;104(35):13857-8. doi: 10.1073/pnas.0706529104. Epub 2007 Aug 20. Proc Natl Acad Sci U S A. 2007. PMID: 17709742 Free PMC article. No abstract available.
Similar articles
-
A potent HIV protease inhibitor, darunavir, does not inhibit ZMPSTE24 or lead to an accumulation of farnesyl-prelamin A in cells.J Biol Chem. 2008 Apr 11;283(15):9797-804. doi: 10.1074/jbc.M709629200. Epub 2008 Jan 28. J Biol Chem. 2008. PMID: 18230615 Free PMC article.
-
HIV-protease inhibitors block the enzymatic activity of purified Ste24p.Biochem Biophys Res Commun. 2008 Sep 19;374(2):365-8. doi: 10.1016/j.bbrc.2008.07.033. Epub 2008 Jul 17. Biochem Biophys Res Commun. 2008. PMID: 18639527 Free PMC article.
-
Inhibitors of protein geranylgeranyltransferase-I lead to prelamin A accumulation in cells by inhibiting ZMPSTE24.J Lipid Res. 2012 Jun;53(6):1176-82. doi: 10.1194/jlr.M026161. Epub 2012 Mar 23. J Lipid Res. 2012. PMID: 22448028 Free PMC article.
-
ZMPSTE24, an integral membrane zinc metalloprotease with a connection to progeroid disorders.Biol Chem. 2009 Aug;390(8):761-73. doi: 10.1515/BC.2009.080. Biol Chem. 2009. PMID: 19453269 Review.
-
Prelamin A, Zmpste24, misshapen cell nuclei, and progeria--new evidence suggesting that protein farnesylation could be important for disease pathogenesis.J Lipid Res. 2005 Dec;46(12):2531-58. doi: 10.1194/jlr.R500011-JLR200. Epub 2005 Oct 5. J Lipid Res. 2005. PMID: 16207929 Review.
Cited by
-
Direct synthesis of lamin A, bypassing prelamin a processing, causes misshapen nuclei in fibroblasts but no detectable pathology in mice.J Biol Chem. 2010 Jul 2;285(27):20818-26. doi: 10.1074/jbc.M110.128835. Epub 2010 May 3. J Biol Chem. 2010. PMID: 20439468 Free PMC article.
-
Biogenesis of the Saccharomyces cerevisiae pheromone a-factor, from yeast mating to human disease.Microbiol Mol Biol Rev. 2012 Sep;76(3):626-51. doi: 10.1128/MMBR.00010-12. Microbiol Mol Biol Rev. 2012. PMID: 22933563 Free PMC article. Review.
-
The farnesyl transferase inhibitor (FTI) lonafarnib improves nuclear morphology in ZMPSTE24-deficient fibroblasts from patients with the progeroid disorder MAD-B.Nucleus. 2023 Dec;14(1):2288476. doi: 10.1080/19491034.2023.2288476. Epub 2023 Dec 5. Nucleus. 2023. PMID: 38050983 Free PMC article.
-
Photoswitchable Isoprenoid Lipids Enable Optical Control of Peptide Lipidation.ACS Chem Biol. 2022 Oct 21;17(10):2945-2953. doi: 10.1021/acschembio.2c00645. Epub 2022 Oct 4. ACS Chem Biol. 2022. PMID: 36194691 Free PMC article.
-
Dynamics of lamin-A processing following precursor accumulation.PLoS One. 2010 May 28;5(5):e10874. doi: 10.1371/journal.pone.0010874. PLoS One. 2010. PMID: 20526372 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Molecular Biology Databases
Research Materials
Miscellaneous

