Sox17 dependence distinguishes the transcriptional regulation of fetal from adult hematopoietic stem cells
- PMID: 17655922
- PMCID: PMC2577201
- DOI: 10.1016/j.cell.2007.06.011
Sox17 dependence distinguishes the transcriptional regulation of fetal from adult hematopoietic stem cells
Abstract
Fetal stem cells differ phenotypically and functionally from adult stem cells in diverse tissues. However, little is known about how these differences are regulated. To address this we compared the gene expression profiles of fetal versus adult hematopoietic stem cells (HSCs) and discovered that the Sox17 transcriptional regulator is specifically expressed in fetal and neonatal but not adult HSCs. Germline deletion of Sox17 led to severe fetal hematopoietic defects, including a lack of detectable definitive HSCs. Conditional deletion of Sox17 from hematopoietic cells led to the loss of fetal and neonatal but not adult HSCs. HSCs stopped expressing Sox17 approximately 4 weeks after birth. During this transition, loss of Sox17 expression correlated with slower proliferation and the acquisition of an adult phenotype by individual HSCs. Sox17 is thus required for the maintenance of fetal and neonatal HSCs and distinguishes their transcriptional regulation from adult HSCs.
Figures
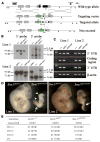




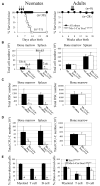

Comment in
-
Fetal to adult stem cell transition: knocking Sox17 off.Cell. 2007 Aug 10;130(3):403-4. doi: 10.1016/j.cell.2007.07.027. Cell. 2007. PMID: 17693249 Review.
References
-
- Azcoitia V, Aracil M, Martinez AC, Torres M. The homeodomain protein Meis1 is essential for definitive hematopoiesis and vascular patterning in the mouse embryo. Dev Biol. 2005;280:307–320. - PubMed
-
- Chambers I, Colby D, Robertson M, Nichols J, Lee S, Tweedie S, Smith A. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell. 2003;113:643–655. - PubMed
-
- Davidson AJ, Ernst P, Wang Y, Dekens MP, Kingsley PD, Palis J, Korsmeyer SJ, Daley GQ, Zon LI. cdx4 mutants fail to specify blood progenitors and can be rescued by multiple hox genes. Nature. 2003;425:300–306. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

