Antagonism of neurosteroid modulation of native gamma-aminobutyric acid receptors by (3alpha,5alpha)-17-phenylandrost-16-en-3-ol
- PMID: 17658511
- PMCID: PMC2098702
- DOI: 10.1016/j.ejphar.2007.06.028
Antagonism of neurosteroid modulation of native gamma-aminobutyric acid receptors by (3alpha,5alpha)-17-phenylandrost-16-en-3-ol
Abstract
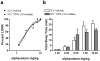
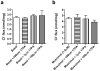
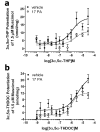
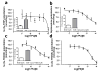
Endogenous pregnane neurosteroids are allosteric modulators at gamma-aminobutyric acid type-A (GABAA) receptors at nanomolar concentrations. There is direct evidence for multiple distinct neurosteroid binding sites on GABAA receptors, dependent upon subunit composition and stoichiometry. This view is supported by the biphasic kinetics of various neuroactive steroids, enantioselectivity of some neurosteroids, selective mutation studies of recombinantly expressed receptors and the selectivity of the neurosteroid antagonist (3alpha,5alpha)-17-phenylandrost-16-en-3-ol (17PA) on 5alpha-pregnane steroid effects on recombinant GABAA receptors expressed in Xenopus oocytes and native receptors in dissociated neurons. However, it is unclear whether this antagonist action is present in a mature mammalian system. The present study evaluated the antagonist activity of 17PA on neurosteroid agonists both in vivo and in vitro by examining the effects of 17PA on 5alpha-pregnane-induced sedation in rats, native mature GABAA receptor ion channels utilizing the chloride flux assay and further studies in recombinant alpha1beta2gamma2 receptors. The data show that 17PA preferentially inhibits 3alpha,5alpha-THP vs. alphaxalone in vivo, preferentially inhibits 3alpha,5alpha-THDOC vs. alphaxalone potentiation of GABA-mediated Cl- uptake in adult cerebral cortical synaptoneurosomes, but shows no specificity for 3alpha,5alpha-THDOC vs. alphaxalone in recombinant alpha1beta2gamma2 receptors. These data provide further evidence of the specificity of 17PA and the heterogeneity of neurosteroid recognition sites on GABAA receptors in the CNS.
Figures
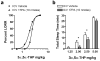

Similar articles
-
Selective antagonism of 5alpha-reduced neurosteroid effects at GABA(A) receptors.Mol Pharmacol. 2004 May;65(5):1191-7. doi: 10.1124/mol.65.5.1191. Mol Pharmacol. 2004. PMID: 15102947
-
Modulation of GABA(A) receptor function by neuroactive steroids: evidence for heterogeneity of steroid sensitivity of recombinant GABA(A) receptor isoforms.Can J Physiol Pharmacol. 1998 Sep;76(9):909-20. doi: 10.1139/cjpp-76-9-909. Can J Physiol Pharmacol. 1998. PMID: 10066142
-
Partial agonism by 3alpha,21-dihydroxy-5beta-pregnan-20-one at the gamma-aminobutyric acidA receptor neurosteroid site.J Pharmacol Exp Ther. 1997 Jun;281(3):1095-101. J Pharmacol Exp Ther. 1997. PMID: 9190841
-
Neurosteroidogenesis: relevance to neurosteroid actions in brain and modulation by psychotropic drugs.Crit Rev Neurobiol. 2004;16(1-2):67-74. doi: 10.1615/critrevneurobiol.v16.i12.70. Crit Rev Neurobiol. 2004. PMID: 15581401 Review.
-
GABAA receptor modulating steroid antagonists (GAMSA) are functional in vivo.J Steroid Biochem Mol Biol. 2016 Jun;160:98-105. doi: 10.1016/j.jsbmb.2015.10.019. Epub 2015 Oct 30. J Steroid Biochem Mol Biol. 2016. PMID: 26523675 Review.
Cited by
-
Allopregnanolone in the bed nucleus of the stria terminalis modulates contextual fear in rats.Front Behav Neurosci. 2015 Aug 4;9:205. doi: 10.3389/fnbeh.2015.00205. eCollection 2015. Front Behav Neurosci. 2015. PMID: 26300750 Free PMC article.
-
Neurosteroid interactions with synaptic and extrasynaptic GABA(A) receptors: regulation of subunit plasticity, phasic and tonic inhibition, and neuronal network excitability.Psychopharmacology (Berl). 2013 Nov;230(2):151-88. doi: 10.1007/s00213-013-3276-5. Epub 2013 Sep 27. Psychopharmacology (Berl). 2013. PMID: 24071826 Free PMC article. Review.
-
Neurosteroids and their potential as a safer class of general anesthetics.J Anesth. 2024 Apr;38(2):261-274. doi: 10.1007/s00540-023-03291-4. Epub 2024 Jan 22. J Anesth. 2024. PMID: 38252143 Free PMC article. Review.
-
Photoaffinity labeling identifies an intersubunit steroid-binding site in heteromeric GABA type A (GABAA) receptors.J Biol Chem. 2020 Aug 14;295(33):11495-11512. doi: 10.1074/jbc.RA120.013452. Epub 2020 Jun 15. J Biol Chem. 2020. PMID: 32540960 Free PMC article.
-
Pleiotropic actions of allopregnanolone underlie therapeutic benefits in stress-related disease.Neurobiol Stress. 2019 Nov 27;12:100203. doi: 10.1016/j.ynstr.2019.100203. eCollection 2020 May. Neurobiol Stress. 2019. PMID: 31879693 Free PMC article.
References
-
- Belelli D, Gee KW. 5α-Pregnan-3α,20α-diol behaves like a partial agonist in the modulation of GABA-stimulated chloride ion uptake by synaptoneurosomes. European Journal of Pharmacology. 1989;167:173–176. - PubMed
-
- Belelli D, Lambert JJ. Neurosteroids: endogenous regulators of the GABA(A) receptor. Nat Rev Neurosci. 2005;6:565–575. - PubMed
-
- Bovolin P, Santi M-R, Memo M, Costa E, Grayson DR. Distinct developmental patterns of rat α1, α5, γ2S and γ2L γ-aminobutyric acid receptor subunit mRNAs in vivo and in vitro. Journal of Neurochemistry. 1992;59:62–72. - PubMed
-
- Callachan H, Cottrell GA, Hather NY, Lambert JJ, Nooney JM, Peters JA. Modulation of the GABAA receptor by progesterone metabolites. Proceedings of the Royal Society of London. Series B: Biological Sciences. 1987;231:359–369. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

