Anaerobic regulation of Shigella flexneri virulence: ArcA regulates Fur and iron acquisition genes
- PMID: 17660284
- PMCID: PMC2045222
- DOI: 10.1128/JB.00621-07
Anaerobic regulation of Shigella flexneri virulence: ArcA regulates Fur and iron acquisition genes
Abstract
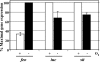
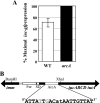
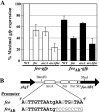
Invasion and plaque formation in epithelial monolayers are routinely used to assess the virulence of Shigella flexneri, a causative agent of dysentery. A modified plaque assay was developed to identify factors contributing to the virulence of S. flexneri under the anaerobic conditions present in the colon. This assay demonstrated the importance of the ferrous iron transport system Feo, as well as the global transcription factors Fur, ArcA, and Fnr, for Shigella plaque formation in anoxic environments. Transcriptional analyses of S. flexneri iron transport genes indicated that anaerobic conditions activated feoABC while repressing genes encoding two other iron transport systems, the ABC transporter Sit and the Iuc/Iut aerobactin siderophore synthesis and transport system. The anaerobic transcription factors ArcA and Fnr activated expression of feoABC, while ArcA repressed iucABCD iutA. Transcription of fur, encoding the iron-responsive transcriptional repressor of bacterial iron acquisition, was also repressed anaerobically in an ArcA-dependent manner.
Figures

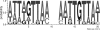

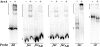
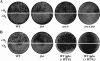
Comment in
-
Environmental stress regulates Shigella virulence: interplay between anerobiosis and iron acquisition.Future Microbiol. 2007 Dec;2(6):601-4. doi: 10.2217/17460913.2.6.601. Future Microbiol. 2007. PMID: 18041901 No abstract available.
Similar articles
-
Contribution of the Shigella flexneri Sit, Iuc, and Feo iron acquisition systems to iron acquisition in vitro and in cultured cells.Infect Immun. 2003 Apr;71(4):1919-28. doi: 10.1128/IAI.71.4.1919-1928.2003. Infect Immun. 2003. PMID: 12654809 Free PMC article.
-
Genetics and environmental regulation of Shigella iron transport systems.Biometals. 2009 Feb;22(1):43-51. doi: 10.1007/s10534-008-9188-x. Epub 2009 Jan 7. Biometals. 2009. PMID: 19130265 Free PMC article.
-
Characterization of ferric and ferrous iron transport systems in Vibrio cholerae.J Bacteriol. 2006 Sep;188(18):6515-23. doi: 10.1128/JB.00626-06. J Bacteriol. 2006. PMID: 16952942 Free PMC article.
-
Iron and pathogenesis of Shigella: iron acquisition in the intracellular environment.Biometals. 2006 Apr;19(2):173-80. doi: 10.1007/s10534-005-4577-x. Biometals. 2006. PMID: 16718602 Review.
-
Shigella flexneri: genetics of entry and intercellular dissemination in epithelial cells.Curr Top Microbiol Immunol. 1994;192:217-41. doi: 10.1007/978-3-642-78624-2_10. Curr Top Microbiol Immunol. 1994. PMID: 7859507 Review. No abstract available.
Cited by
-
Transcriptional regulation of bacterial virulence gene expression by molecular oxygen and nitric oxide.Virulence. 2014;5(8):794-809. doi: 10.4161/viru.27794. Epub 2014 Oct 31. Virulence. 2014. PMID: 25603427 Free PMC article. Review.
-
Transcriptional and functional analysis of the Neisseria gonorrhoeae Fur regulon.J Bacteriol. 2010 Jan;192(1):77-85. doi: 10.1128/JB.00741-09. J Bacteriol. 2010. PMID: 19854902 Free PMC article.
-
Iron, copper, zinc, and manganese transport and regulation in pathogenic Enterobacteria: correlations between strains, site of infection and the relative importance of the different metal transport systems for virulence.Front Cell Infect Microbiol. 2013 Dec 5;3:90. doi: 10.3389/fcimb.2013.00090. eCollection 2013. Front Cell Infect Microbiol. 2013. PMID: 24367764 Free PMC article. Review.
-
Pseudomonas aeruginosa uses multiple pathways to acquire iron during chronic infection in cystic fibrosis lungs.Infect Immun. 2013 Aug;81(8):2697-704. doi: 10.1128/IAI.00418-13. Epub 2013 May 20. Infect Immun. 2013. PMID: 23690396 Free PMC article.
-
Environmental factors affecting the expression of pilAB as well as the proteome and transcriptome of the grass endophyte Azoarcus sp. strain BH72.PLoS One. 2012;7(1):e30421. doi: 10.1371/journal.pone.0030421. Epub 2012 Jan 20. PLoS One. 2012. PMID: 22276194 Free PMC article.
References
-
- Ausubel, F. M., R. Brent, R. E. Kingston, D. D. Moore, J. G. Seidman, J. A. Smith, and K. Struhl. 1999. Short protocols in molecular biology: a compendium of methods from Current Protocols in Molecular Biology, 4th ed. Wiley, New York, NY.
-
- Chiancone, E., P. Ceci, A. Ilari, F. Ribacchi, and S. Stefanini. 2004. Iron and proteins for iron storage and detoxification. Biometals 17:197-202. - PubMed
-
- Cianciotto, N. P. 2007. Iron acquisition by Legionella pneumophila. Biometals 20:323-331. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

