A bacterial transgene for catalase protects translation of d1 protein during exposure of salt-stressed tobacco leaves to strong light
- PMID: 17660354
- PMCID: PMC1976566
- DOI: 10.1104/pp.107.101733
A bacterial transgene for catalase protects translation of d1 protein during exposure of salt-stressed tobacco leaves to strong light
Abstract
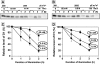
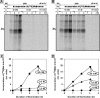
During photoinhibition of photosystem II (PSII) in cyanobacteria, salt stress inhibits the repair of photodamaged PSII and, in particular, the synthesis of the D1 protein (D1). We investigated the effects of salt stress on the repair of PSII and the synthesis of D1 in wild-type tobacco (Nicotiana tabacum 'Xanthi') and in transformed plants that harbored the katE gene for catalase from Escherichia coli. Salt stress due to NaCl enhanced the photoinhibition of PSII in leaf discs from both wild-type and katE-transformed plants, but the effect of salt stress was less significant in the transformed plants than in wild-type plants. In the presence of lincomycin, which inhibits protein synthesis in chloroplasts, the activity of PSII decreased rapidly and at similar rates in both types of leaf disc during photoinhibition, and the observation suggests that repair of PSII was protected by the transgene-coded enzyme. Incorporation of [(35)S]methionine into D1 during photoinhibition was inhibited by salt stress, and the transformation mitigated this inhibitory effect. Northern blotting revealed that the level of psbA transcripts was not significantly affected by salt stress or by the transformation. Our results suggest that salt stress enhanced photoinhibition by inhibiting repair of PSII and that the katE transgene increased the resistance of the chloroplast's translational machinery to salt stress by scavenging hydrogen peroxide.
Figures



Similar articles
-
Glycinebetaine counteracts the inhibitory effects of salt stress on the degradation and synthesis of D1 protein during photoinhibition in Synechococcus sp. PCC 7942.Plant Physiol. 2006 Jun;141(2):758-65. doi: 10.1104/pp.106.076976. Epub 2006 Apr 21. Plant Physiol. 2006. PMID: 16632587 Free PMC article.
-
Differential turnover of the photosystem II reaction centre D1 protein in mesophyll and bundle sheath chloroplasts of maize.Biochim Biophys Acta. 2009 Oct;1787(10):1161-9. doi: 10.1016/j.bbabio.2009.05.002. Epub 2009 May 18. Biochim Biophys Acta. 2009. PMID: 19450540
-
Mitochondrial alternative oxidase pathway protects plants against photoinhibition by alleviating inhibition of the repair of photodamaged PSII through preventing formation of reactive oxygen species in Rumex K-1 leaves.Physiol Plant. 2011 Dec;143(4):396-407. doi: 10.1111/j.1399-3054.2011.01514.x. Epub 2011 Oct 3. Physiol Plant. 2011. PMID: 21883255
-
Protein synthesis is the primary target of reactive oxygen species in the photoinhibition of photosystem II.Physiol Plant. 2011 May;142(1):35-46. doi: 10.1111/j.1399-3054.2011.01457.x. Epub 2011 Mar 2. Physiol Plant. 2011. PMID: 21320129 Review.
-
A new paradigm for the action of reactive oxygen species in the photoinhibition of photosystem II.Biochim Biophys Acta. 2006 Jul;1757(7):742-9. doi: 10.1016/j.bbabio.2006.05.013. Epub 2006 May 23. Biochim Biophys Acta. 2006. PMID: 16784721 Review.
Cited by
-
Understanding the roles of osmolytes for acclimatizing plants to changing environment: a review of potential mechanism.Plant Signal Behav. 2021 Aug 3;16(8):1913306. doi: 10.1080/15592324.2021.1913306. Epub 2021 Jun 16. Plant Signal Behav. 2021. PMID: 34134596 Free PMC article. Review.
-
Bioengineering for salinity tolerance in plants: state of the art.Mol Biotechnol. 2013 May;54(1):102-23. doi: 10.1007/s12033-012-9538-3. Mol Biotechnol. 2013. PMID: 22539206 Review.
-
Structural and functional changes of PSI-LHCI supercomplexes of Chlamydomonas reinhardtii cells grown under high salt conditions.Planta. 2010 Mar;231(4):913-22. doi: 10.1007/s00425-009-1097-x. Planta. 2010. PMID: 20183922
-
Ecological genomics of marine picocyanobacteria.Microbiol Mol Biol Rev. 2009 Jun;73(2):249-99. doi: 10.1128/MMBR.00035-08. Microbiol Mol Biol Rev. 2009. PMID: 19487728 Free PMC article. Review.
-
Biopolymers as Seed-Coating Agent to Enhance Microbially Induced Tolerance of Barley to Phytopathogens.Polymers (Basel). 2024 Jan 30;16(3):376. doi: 10.3390/polym16030376. Polymers (Basel). 2024. PMID: 38337265 Free PMC article.
References
-
- Allakhverdiev SI, Murata N (2004) Environmental stress inhibits the synthesis de novo of proteins involved in the photodamage-repair cycle of photosystem II in Synechocystis sp. PCC 6803. Biochim Biophys Acta 1657 23–32 - PubMed
-
- Barber J, Andersson B (1992) Too much of a good thing: light can be bad for photosynthesis. Trends Biochem Sci 17 61–66 - PubMed
-
- Boyer JS (1982) Plant productivity and environment. Science 218 443–448 - PubMed
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

