Astrin is required for the maintenance of sister chromatid cohesion and centrosome integrity
- PMID: 17664331
- PMCID: PMC2064855
- DOI: 10.1083/jcb.200701163
Astrin is required for the maintenance of sister chromatid cohesion and centrosome integrity
Abstract
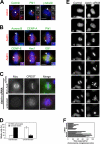
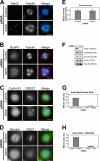
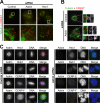
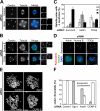
Faithful chromosome segregation in mitosis requires the formation of a bipolar mitotic spindle with stably attached chromosomes. Once all of the chromosomes are aligned, the connection between the sister chromatids is severed by the cysteine protease separase. Separase also promotes centriole disengagement at the end of mitosis. Temporal coordination of these two activities with the rest of the cell cycle is required for the successful completion of mitosis. In this study, we report that depletion of the microtubule and kinetochore protein astrin results in checkpoint-arrested cells with multipolar spindles and separated sister chromatids, which is consistent with untimely separase activation. Supporting this idea, astrin-depleted cells contain active separase, and separase depletion suppresses the premature sister chromatid separation and centriole disengagement in these cells. We suggest that astrin contributes to the regulatory network that controls separase activity.
Figures
Similar articles
-
Centrosomal Aki1 and cohesin function in separase-regulated centriole disengagement.J Cell Biol. 2009 Nov 30;187(5):607-14. doi: 10.1083/jcb.200906019. Epub 2009 Nov 23. J Cell Biol. 2009. PMID: 19948489 Free PMC article.
-
Separase and Roads to Disengage Sister Chromatids during Anaphase.Int J Mol Sci. 2023 Feb 27;24(5):4604. doi: 10.3390/ijms24054604. Int J Mol Sci. 2023. PMID: 36902034 Free PMC article. Review.
-
Recruitment of separase to mitotic chromosomes is regulated by Aurora B.Cell Cycle. 2009 May 1;8(9):1433-43. doi: 10.4161/cc.8.9.8331. Epub 2009 May 22. Cell Cycle. 2009. PMID: 19342897
-
Kendrin is a novel substrate for separase involved in the licensing of centriole duplication.Curr Biol. 2012 May 22;22(10):915-21. doi: 10.1016/j.cub.2012.03.048. Epub 2012 Apr 26. Curr Biol. 2012. PMID: 22542101
-
Sister chromatid tension and the spindle assembly checkpoint.Curr Opin Cell Biol. 2009 Dec;21(6):785-95. doi: 10.1016/j.ceb.2009.09.007. Epub 2009 Oct 19. Curr Opin Cell Biol. 2009. PMID: 19846287 Review.
Cited by
-
Astrin: A Key Player in Mitosis and Cancer.Front Cell Dev Biol. 2020 Aug 28;8:866. doi: 10.3389/fcell.2020.00866. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32984344 Free PMC article. Review.
-
Is cohesin required for spindle-pole-body/centrosome cohesion?Commun Integr Biol. 2012 Jan 1;5(1):26-9. doi: 10.4161/cib.18557. Commun Integr Biol. 2012. PMID: 22482005 Free PMC article.
-
3D-structured illumination microscopy provides novel insight into architecture of human centrosomes.Biol Open. 2012 Oct 15;1(10):965-76. doi: 10.1242/bio.20122337. Epub 2012 Aug 8. Biol Open. 2012. PMID: 23213374 Free PMC article.
-
Aurora-B kinase pathway controls the lateral to end-on conversion of kinetochore-microtubule attachments in human cells.Nat Commun. 2017 Jul 28;8(1):150. doi: 10.1038/s41467-017-00209-z. Nat Commun. 2017. PMID: 28751710 Free PMC article.
-
Cep57, a NEDD1-binding pericentriolar material component, is essential for spindle pole integrity.Cell Res. 2012 Sep;22(9):1390-401. doi: 10.1038/cr.2012.61. Epub 2012 Apr 17. Cell Res. 2012. PMID: 22508265 Free PMC article.
References
-
- Baumann, C., R. Korner, K. Hofmann, and E.A. Nigg. 2007. PICH, a centromere-associated SNF2 family ATPase, is regulated by Plk1 and required for the spindle checkpoint. Cell. 128:101–114. - PubMed
-
- Chang, M.S., C.J. Huang, M.L. Chen, S.T. Chen, C.C. Fan, J.M. Chu, W.C. Lin, and Y.C. Yang. 2001. Cloning and characterization of hMAP126, a new member of mitotic spindle-associated proteins. Biochem. Biophys. Res. Commun. 287:116–121. - PubMed
-
- Ciosk, R., W. Zachariae, C. Michaelis, A. Shevchenko, M. Mann, and K. Nasmyth. 1998. An ESP1/PDS1 complex regulates loss of sister chromatid cohesion at the metaphase to anaphase transition in yeast. Cell. 93:1067–1076. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

