uPAR induces epithelial-mesenchymal transition in hypoxic breast cancer cells
- PMID: 17664334
- PMCID: PMC2064849
- DOI: 10.1083/jcb.200701092
uPAR induces epithelial-mesenchymal transition in hypoxic breast cancer cells
Abstract
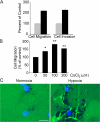
Hypoxia activates genetic programs that facilitate cell survival; however, in cancer, it may promote invasion and metastasis. In this study, we show that breast cancer cells cultured in 1.0% O(2) demonstrate changes consistent with epithelial-mesenchymal transition (EMT). Snail translocates to the nucleus, and E-cadherin is lost from plasma membranes. Vimentin expression, cell migration, Matrigel invasion, and collagen remodeling are increased. Hypoxia-induced EMT is accompanied by increased expression of the urokinase-type plasminogen activator receptor (uPAR) and activation of cell signaling factors downstream of uPAR, including Akt and Rac1. Glycogen synthase kinase-3beta is phosphorylated, and Snail expression is increased. Hypoxia-induced EMT is blocked by uPAR gene silencing and mimicked by uPAR overexpression in normoxia. Antagonizing Rac1 or phosphatidylinositol 3-kinase also inhibits development of cellular properties associated with EMT in hypoxia. Breast cancer cells implanted on chick chorioallantoic membranes and treated with CoCl(2), to model hypoxia, demonstrate increased dissemination. We conclude that in hypoxia, uPAR activates diverse cell signaling pathways that cooperatively induce EMT and may promote cancer metastasis.
Figures









References
-
- Akakura, N., M. Kobayashi, I. Horiuchi, A. Suzuki, J. Wang, J. Chen, H. Niizeki, K. Kawamura, M. Hosokawa, and M. Asaka. 2001. Constitutive expression of hypoxia-inducible factor-1alpha renders pancreatic cancer cells resistant to apoptosis induced by hypoxia and nutrient deprivation. Cancer Res. 61:6548–6554. - PubMed
-
- Alfano, D., I. Iaccarino, and M.P. Stoppelli. 2006. Urokinase signaling through tts receptor protects against anoikis by increasing BCL-xL expression levels. J. Biol. Chem. 281:17758–17767. - PubMed
-
- Anand-Apte, B., B.R. Zetter, A. Viswanathan, R.-G. Qiu, J. Chen, R. Ruggieri, and M. Symons. 1997. Platelet-derived growth factor and fibronectin-stimulated migration are differentially regulated by the Rac and extracellular signal-regulated kinase pathways. J. Biol. Chem. 272:30688–30692. - PubMed
-
- Andreasen, P.A., L. Kjøller, L. Christenson, and M.J. Duffy. 1997. The urokinase- type plasminogen activator system in cancer metastasis: a review. Int. J. Cancer. 72:1–22. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

