Responses of rat P2X2 receptors to ultrashort pulses of ATP provide insights into ATP binding and channel gating
- PMID: 17664346
- PMCID: PMC2151634
- DOI: 10.1085/jgp.200709779
Responses of rat P2X2 receptors to ultrashort pulses of ATP provide insights into ATP binding and channel gating
Abstract
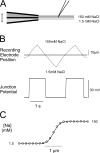
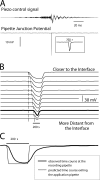
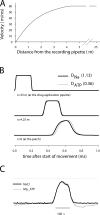
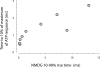
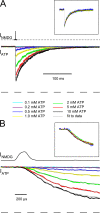
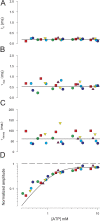
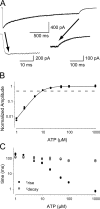
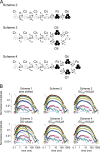
To gain insight into the way that P2X(2) receptors localized at synapses might function, we explored the properties of outside-out patches containing many of these channels as ATP was very rapidly applied and removed. Using a new method to calibrate the speed of exchange of solution over intact patches, we were able to reliably produce applications of ATP lasting <200 micros. For all concentrations of ATP, there was a delay of at least 80 micros between the time when ATP arrived at the receptor and the first detectable flow of inward current. In response to 200-micros pulses of ATP, the time constant of the rising phase of the current was approximately 600 micros. Thus, most channel openings occurred when no free ATP was present. The current deactivated with a time constant of approximately 60 ms. The amplitude of the peak response to a brief pulse of a saturating concentration of ATP was approximately 70% of that obtained during a long application of the same concentration of ATP. Thus, ATP leaves fully liganded channels without producing an opening at least 30% of the time. Extensive kinetic modeling revealed three different schemes that fit the data well, a sequential model and two allosteric models. To account for the delay in opening at saturating ATP, it was necessary to incorporate an intermediate closed state into all three schemes. These kinetic properties indicate that responses to ATP at synapses that use homomeric P2X(2) receptors would be expected to greatly outlast the duration of the synaptic ATP transient produced by a single presynaptic spike. Like NMDA receptors, P2X(2) receptors provide the potential for complex patterns of synaptic integration over a time scale of hundreds of milliseconds.
Figures



Similar articles
-
The single-channel properties of purinergic P2X ATP receptors in outside-out patches from rat hypothalamic paraventricular parvocells.Pflugers Arch. 2001 Oct;443(1):115-22. doi: 10.1007/s004240100624. Pflugers Arch. 2001. PMID: 11692275
-
Single channel properties of P2X2 purinoceptors.J Gen Physiol. 1999 May;113(5):695-720. doi: 10.1085/jgp.113.5.695. J Gen Physiol. 1999. PMID: 10228183 Free PMC article.
-
Single channel properties of P2X ATP receptors in outside-out patches from rat hippocampal granule cells.J Physiol. 2000 Sep 15;527 Pt 3(Pt 3):529-47. doi: 10.1111/j.1469-7793.2000.00529.x. J Physiol. 2000. PMID: 10990539 Free PMC article.
-
Pharmacology of P2X channels.Pflugers Arch. 2006 Aug;452(5):513-37. doi: 10.1007/s00424-006-0070-9. Epub 2006 Apr 29. Pflugers Arch. 2006. PMID: 16649055 Review.
-
Synaptic P2X receptors.Curr Opin Neurobiol. 2001 Jun;11(3):378-86. doi: 10.1016/s0959-4388(00)00222-1. Curr Opin Neurobiol. 2001. PMID: 11399438 Review.
Cited by
-
Dynamics of T-Junction Solution Switching Aimed at Patch Clamp Experiments.PLoS One. 2015 Jul 15;10(7):e0133187. doi: 10.1371/journal.pone.0133187. eCollection 2015. PLoS One. 2015. PMID: 26177538 Free PMC article.
-
The activation mechanism of alpha1beta2gamma2S and alpha3beta3gamma2S GABAA receptors.J Gen Physiol. 2010 Jan;135(1):59-75. doi: 10.1085/jgp.200910317. J Gen Physiol. 2010. PMID: 20038526 Free PMC article.
-
Exploring the ATP-binding site of P2X receptors.Front Cell Neurosci. 2013 Dec 30;7:273. doi: 10.3389/fncel.2013.00273. Front Cell Neurosci. 2013. PMID: 24415999 Free PMC article. Review.
-
Key sites for P2X receptor function and multimerization: overview of mutagenesis studies on a structural basis.Curr Med Chem. 2015;22(7):799-818. doi: 10.2174/0929867322666141128163215. Curr Med Chem. 2015. PMID: 25439586 Free PMC article. Review.
-
Modeling a Nociceptive Neuro-Immune Synapse Activated by ATP and 5-HT in Meninges: Novel Clues on Transduction of Chemical Signals Into Persistent or Rhythmic Neuronal Firing.Front Cell Neurosci. 2020 May 19;14:135. doi: 10.3389/fncel.2020.00135. eCollection 2020. Front Cell Neurosci. 2020. PMID: 32508598 Free PMC article.
References
-
- Banke, T.G., and S.F. Traynelis. 2003. Activation of NR1/NR2B NMDA receptors. Nat. Neurosci. 6:144–152. - PubMed
-
- Barry, P.H., and J.W. Lynch. 1991. Liquid junction potentials and small cell effects in patch-clamp analysis. J. Membr. Biol. 121:101–117. - PubMed
-
- Bartoletti, S., B.D. Flury, and D.G. Nel. 1999. Allometric extension. Biometrics. 55:1210–1214. - PubMed
-
- Brooks, S.P., and K.B. Storey. 1992. Bound and determined: a computer program for making buffers of defined ion concentrations. Anal. Biochem. 201:119–126. - PubMed

