A case study on the effect of neutralizing antibodies to interferon beta 1b in multiple sclerosis patients followed for 3 years with monthly imaging
- PMID: 17666095
- PMCID: PMC2219272
- DOI: 10.1111/j.1365-2249.2007.03467.x
A case study on the effect of neutralizing antibodies to interferon beta 1b in multiple sclerosis patients followed for 3 years with monthly imaging
Abstract
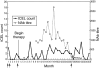
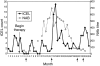
Interferon beta (IFN-beta) is among the first-line treatment options for patients with multiple sclerosis (MS). A potential caveat of therapy, however, is the development of neutralizing antibodies (NAb) and/or neutralizing activity (NA) non-antibody mediated, although debate is still ongoing as to whether NAb significantly hampers the efficacy of the drug or rather represents an immunologically irrelevant epiphenomenon. In the present study, we describe the effect of NAb on IFN-beta-1b through clinical and magnetic resonance imaging (MRI) outcome measures of five relapsing-remitting multiple sclerosis (RRMS) patients who were treated with 250 mug of subcutaneously administered IFN-beta-1b every other day and developed NAb at varying titres and times during the course of therapy. Despite the small number of NAb(+) patients, heterogeneity in MRI/clinical response to IFN-beta-1b was identified. Response to IFN-beta-1b therapy was observed in the absence or presence of NAb. Also observed was failure to IFN-beta-1b coincident with high and sustained NAb titres, but also before NAb development or in the presence of low NAb titres. Multiple MRI and NAb measurements performed within the same individual allow for a better description of the complex heterogeneous response to IFN-beta-1b with respect to NAb occurrence.
Figures



Similar articles
-
The clinical effect of neutralizing antibodies against interferon-beta is independent of the type of interferon-beta used for patients with relapsing-remitting multiple sclerosis.Mult Scler. 2009 May;15(5):601-5. doi: 10.1177/1352458508101946. Epub 2009 Mar 19. Mult Scler. 2009. PMID: 19299439
-
MRI activity and neutralising antibody as predictors of response to interferon beta treatment in multiple sclerosis.J Neurol Neurosurg Psychiatry. 2008 Jun;79(6):646-51. doi: 10.1136/jnnp.2007.130229. Epub 2007 Nov 6. J Neurol Neurosurg Psychiatry. 2008. PMID: 17986500 Clinical Trial.
-
Neutralizing antibodies to interferon beta-1b multiple sclerosis: a clinico-radiographic paradox in the BEYOND trial.Mult Scler. 2012 Feb;18(2):181-95. doi: 10.1177/1352458511418629. Epub 2011 Sep 27. Mult Scler. 2012. PMID: 21952094 Clinical Trial.
-
Guidelines on use of anti-IFN-beta antibody measurements in multiple sclerosis: report of an EFNS Task Force on IFN-beta antibodies in multiple sclerosis.Eur J Neurol. 2005 Nov;12(11):817-27. doi: 10.1111/j.1468-1331.2005.01386.x. Eur J Neurol. 2005. PMID: 16241970 Review.
-
The clinical importance of neutralizing antibodies in relapsing-remitting multiple sclerosis.Curr Med Res Opin. 2006 Feb;22(2):223-39. doi: 10.1185/030079906X80413. Curr Med Res Opin. 2006. PMID: 16466595 Review.
Cited by
-
MxA mRNA quantification and disability progression in interferon beta-treated multiple sclerosis patients.PLoS One. 2014 Apr 14;9(4):e94794. doi: 10.1371/journal.pone.0094794. eCollection 2014. PLoS One. 2014. PMID: 24733382 Free PMC article.
-
Neutralizing antibodies against interferon-Beta.Ther Adv Neurol Disord. 2008 Sep;1(2):125-41. doi: 10.1177/1756285608095144. Ther Adv Neurol Disord. 2008. PMID: 21180570 Free PMC article.
-
Ring and nodular multiple sclerosis lesions: a retrospective natural history study.Neurology. 2010 Mar 9;74(10):851-6. doi: 10.1212/WNL.0b013e3181d31df5. Neurology. 2010. PMID: 20211910 Free PMC article.
-
A Population Approach to Characterize Interferon Beta-1b Effect on Contrast Enhancing Lesions in Patients With Relapsing Remitting Multiple Sclerosis.CPT Pharmacometrics Syst Pharmacol. 2015 May;4(5):295-304. doi: 10.1002/psp4.36. Epub 2015 Apr 24. CPT Pharmacometrics Syst Pharmacol. 2015. PMID: 26225255 Free PMC article.
-
Development of interferon beta-neutralising antibodies in multiple sclerosis--a systematic review and meta-analysis.Eur J Clin Pharmacol. 2015 Nov;71(11):1287-98. doi: 10.1007/s00228-015-1921-0. Epub 2015 Aug 14. Eur J Clin Pharmacol. 2015. PMID: 26268445
References
-
- IFNB Multiple Sclerosis Study Group and the University of British Columbia MS/MRI Analysis Group. Neutralizing antibodies during treatment of multiple sclerosis with interferon beta-1b: experience during the first three years. Neurology. 1996;47:889–94. - PubMed
-
- Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS) Neurology. 1983;33:1444–52. - PubMed
-
- Bagnato F, Pozzilli C. Pharmacological methods to overcome IFN-beta antibody formation in the treatment of multiple sclerosis. Expert Opin Invest Drugs. 2003;7:1153–63. - PubMed
-
- Sorensen PS, Deisenhammer F, Duda P, et al. Guidelines on use of anti-IFN-β antibodies in multiple sclerosis: a report of an EFNS Task Force on IFN-β in multiple sclerosis. Eur J Neurol. 2005;12:817–27. - PubMed
-
- Namaka M, Pollitt-Smith M, Gupta A, et al. The clinical importance of neutralizing antibodies in relapsing–remitting multiple sclerosis. Curr Med Res Opin. 2006;22:223–39. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

