Faithful segregation of the multicopy yeast plasmid through cohesin-mediated recognition of sisters
- PMID: 17670945
- PMCID: PMC1941829
- DOI: 10.1073/pnas.0702996104
Faithful segregation of the multicopy yeast plasmid through cohesin-mediated recognition of sisters
Abstract
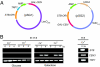
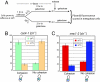
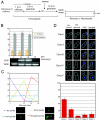
The 2-microm yeast plasmid, a benign high-copy nuclear parasite, propagates itself with nearly the same fidelity as the chromosomes of its host. Equal plasmid segregation is absolutely dependent on the cohesin complex assembled at the plasmid partitioning locus STB. However, the mechanism of cohesin action in the context of multiple plasmid copies, resident within two separate clusters after DNA replication, is unknown. By using "single-copy" derivatives of the 2-microm plasmid, we demonstrate that recruitment of cohesin at STB during S phase indeed translates into cohesion between plasmid molecules. Through binary fluorescence tagging, we reveal that segregation of replicated plasmids occurs in a sister-to-sister fashion. Thus, cohesin serves the same fundamental purpose in plasmid and chromosome segregation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
A novel role for the mitotic spindle during DNA segregation in yeast: promoting 2 microm plasmid-cohesin association.Mol Cell Biol. 2005 May;25(10):4283-98. doi: 10.1128/MCB.25.10.4283-4298.2005. Mol Cell Biol. 2005. PMID: 15870297 Free PMC article.
-
Mutations in a partitioning protein and altered chromatin structure at the partitioning locus prevent cohesin recruitment by the Saccharomyces cerevisiae plasmid and cause plasmid missegregation.Mol Cell Biol. 2004 Jun;24(12):5290-303. doi: 10.1128/MCB.24.12.5290-5303.2004. Mol Cell Biol. 2004. PMID: 15169893 Free PMC article.
-
The centromere-specific histone variant Cse4p (CENP-A) is essential for functional chromatin architecture at the yeast 2-microm circle partitioning locus and promotes equal plasmid segregation.J Cell Biol. 2006 Sep 11;174(6):779-90. doi: 10.1083/jcb.200603042. J Cell Biol. 2006. PMID: 16966420 Free PMC article.
-
Sister chromatid cohesion: the cohesin cleavage model does not ring true.Genes Cells. 2007 Jun;12(6):693-708. doi: 10.1111/j.1365-2443.2007.01093.x. Genes Cells. 2007. PMID: 17573771 Review.
-
Segregation of the yeast plasmid: similarities and contrasts with bacterial plasmid partitioning.Plasmid. 2004 May;51(3):162-78. doi: 10.1016/j.plasmid.2004.02.005. Plasmid. 2004. PMID: 15109823 Review.
Cited by
-
Temporal sequence and cell cycle cues in the assembly of host factors at the yeast 2 micron plasmid partitioning locus.Nucleic Acids Res. 2013 Feb 1;41(4):2340-53. doi: 10.1093/nar/gks1338. Epub 2012 Dec 28. Nucleic Acids Res. 2013. PMID: 23275556 Free PMC article.
-
Yeast cohesin complex embraces 2 micron plasmid sisters in a tri-linked catenane complex.Nucleic Acids Res. 2010 Jan;38(2):570-84. doi: 10.1093/nar/gkp993. Epub 2009 Nov 17. Nucleic Acids Res. 2010. PMID: 19920123 Free PMC article.
-
The selfish yeast plasmid uses the nuclear motor Kip1p but not Cin8p for its localization and equal segregation.J Cell Biol. 2009 Apr 20;185(2):251-64. doi: 10.1083/jcb.200810130. Epub 2009 Apr 13. J Cell Biol. 2009. PMID: 19364922 Free PMC article.
-
Histone H3-variant Cse4-induced positive DNA supercoiling in the yeast plasmid has implications for a plasmid origin of a chromosome centromere.Proc Natl Acad Sci U S A. 2011 Aug 16;108(33):13671-6. doi: 10.1073/pnas.1101944108. Epub 2011 Aug 1. Proc Natl Acad Sci U S A. 2011. PMID: 21807992 Free PMC article.
-
Cse4 (CenH3) association with the Saccharomyces cerevisiae plasmid partitioning locus in its native and chromosomally integrated states: implications in centromere evolution.Mol Cell Biol. 2011 Mar;31(5):1030-40. doi: 10.1128/MCB.01191-10. Epub 2010 Dec 20. Mol Cell Biol. 2011. PMID: 21173161 Free PMC article.
References
-
- Ghosh SK, Hajra S, Paek A, Jayaram M. Annu Rev Biochem. 2006;75:211–241. - PubMed
-
- Jayaram M, Mehta S, Uzri D, Velmurugan S. Plasmid. 2004;51:162–178. - PubMed
-
- Meluh PB, Yang P, Glowczewski L, Koshland D, Smith MM. Cell. 1998;94:607–613. - PubMed
-
- Huang J, Hsu JM, Laurent BC. Mol Cell. 2004;13:739–750. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

