Foxo3 is required for the regulation of oxidative stress in erythropoiesis
- PMID: 17671650
- PMCID: PMC1934587
- DOI: 10.1172/JCI31807
Foxo3 is required for the regulation of oxidative stress in erythropoiesis
Abstract
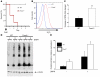
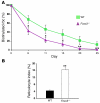
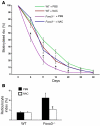
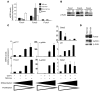
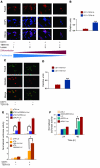
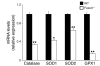
Erythroid cells accumulate hemoglobin as they mature and as a result are highly prone to oxidative damage. However, mechanisms of transcriptional control of antioxidant defense in erythroid cells have thus far been poorly characterized. We observed that animals deficient in the forkhead box O3 (Foxo3) transcription factor died rapidly when exposed to erythroid oxidative stress-induced conditions, while wild-type mice showed no decreased viability. In view of this striking finding, we investigated the potential role of Foxo3 in the regulation of ROS in erythropoiesis. Foxo3 expression, nuclear localization, and transcriptional activity were all enhanced during normal erythroid cell maturation. Foxo3-deficient erythrocytes exhibited decreased expression of ROS scavenging enzymes and had a ROS-mediated shortened lifespan and evidence of oxidative damage. Furthermore, loss of Foxo3 induced mitotic arrest in erythroid precursor cells, leading to a significant decrease in the rate of in vivo erythroid maturation. We identified ROS-mediated upregulation of p21(CIP1/WAF1/Sdi1) (also known as Cdkn1a) as a major contributor to the interference with cell cycle progression in Foxo3-deficient erythroid precursor cells. These findings establish an essential nonredundant function for Foxo3 in the regulation of oxidative stress, cell cycle, maturation, and lifespan of erythroid cells. These results may have an impact on the understanding of human disorders in which ROS play a role.
Figures





Comment in
-
Regulation of erythrocyte lifespan: do reactive oxygen species set the clock?J Clin Invest. 2007 Aug;117(8):2075-7. doi: 10.1172/JCI32559. J Clin Invest. 2007. PMID: 17671642 Free PMC article.
Similar articles
-
Regulation of erythrocyte lifespan: do reactive oxygen species set the clock?J Clin Invest. 2007 Aug;117(8):2075-7. doi: 10.1172/JCI32559. J Clin Invest. 2007. PMID: 17671642 Free PMC article.
-
Foxo3 is essential for the regulation of ataxia telangiectasia mutated and oxidative stress-mediated homeostasis of hematopoietic stem cells.J Biol Chem. 2008 Sep 12;283(37):25692-25705. doi: 10.1074/jbc.M800517200. Epub 2008 Apr 18. J Biol Chem. 2008. PMID: 18424439
-
miR-9 upregulation leads to inhibition of erythropoiesis by repressing FoxO3.Sci Rep. 2018 Apr 25;8(1):6519. doi: 10.1038/s41598-018-24628-0. Sci Rep. 2018. PMID: 29695725 Free PMC article.
-
Oxidative stress in the regulation of normal and neoplastic hematopoiesis.Antioxid Redox Signal. 2008 Nov;10(11):1923-40. doi: 10.1089/ars.2008.2142. Antioxid Redox Signal. 2008. PMID: 18707226 Free PMC article. Review.
-
FoxO3 and oxidative stress: a multifaceted role in cellular adaptation.J Mol Med (Berl). 2023 Feb;101(1-2):83-99. doi: 10.1007/s00109-022-02281-5. Epub 2023 Jan 4. J Mol Med (Berl). 2023. PMID: 36598531 Review.
Cited by
-
Role of TRPM2 in cell proliferation and susceptibility to oxidative stress.Am J Physiol Cell Physiol. 2013 Mar;304(6):C548-60. doi: 10.1152/ajpcell.00069.2012. Epub 2013 Jan 9. Am J Physiol Cell Physiol. 2013. PMID: 23302782 Free PMC article.
-
Cellular and Molecular Mechanisms of Environmental Pollutants on Hematopoiesis.Int J Mol Sci. 2020 Sep 23;21(19):6996. doi: 10.3390/ijms21196996. Int J Mol Sci. 2020. PMID: 32977499 Free PMC article. Review.
-
Advances in understanding the mechanisms of erythropoiesis in homeostasis and disease.Br J Haematol. 2016 Sep;174(5):661-73. doi: 10.1111/bjh.14194. Epub 2016 Jul 21. Br J Haematol. 2016. PMID: 27442953 Free PMC article. Review.
-
Metformin as a Fetal Hemoglobin Inducer in Non-transfusion Dependent Thalassemia Patients.Indian J Hematol Blood Transfus. 2024 Jan;40(1):68-73. doi: 10.1007/s12288-023-01662-1. Epub 2023 May 5. Indian J Hematol Blood Transfus. 2024. PMID: 38312174 Free PMC article.
-
From estrogen-centric to aging and oxidative stress: a revised perspective of the pathogenesis of osteoporosis.Endocr Rev. 2010 Jun;31(3):266-300. doi: 10.1210/er.2009-0024. Epub 2010 Jan 5. Endocr Rev. 2010. PMID: 20051526 Free PMC article. Review.
References
-
- Beckman K.B., Ames B.N. The free radical theory of aging matures. Physiol. Rev. 1998;78:547–581. - PubMed
-
- Ito K., et al. Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature. 2004;431:997–1002. - PubMed
-
- Cumming R.C., et al. Fanconi anemia group C protein prevents apoptosis in hematopoietic cells through redox regulation of GSTP1. Nat. Med. 2001;7:814–820. - PubMed
-
- Lin K., Dorman J.B., Rodan A., Kenyon C. daf-16: An HNF-3/forkhead family member that can function to double the life-span of Caenorhabditis elegans. Science. 1997;278:1319–1322. - PubMed
-
- Lehtinen M.K., et al. A conserved MST-FOXO signaling pathway mediates oxidative-stress responses and extends life span. Cell. 2006;125:987–1001. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

