The melanocortinergic pathway is rapidly recruited by emotional stress and contributes to stress-induced anorexia and anxiety-like behavior
- PMID: 17673512
- PMCID: PMC3708592
- DOI: 10.1210/en.2007-0745
The melanocortinergic pathway is rapidly recruited by emotional stress and contributes to stress-induced anorexia and anxiety-like behavior
Abstract
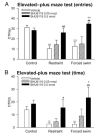
Neurons producing melanocortin receptor agonist, alpha-MSH derived from proopiomelanocortin, and antagonist, agouti-related protein, are known to be sensitive to metabolic stress such as food deprivation and glucoprivation. However, how these neurons respond to emotional/psychological stress remained to be elucidated. We report here that acute emotional stressors, i.e. restraint and forced swim, evoked mRNA expression of c-fos, a neuronal activation marker, in a high percentage of proopiomelanocortin neurons (up to 53% for restraint stress and 62% for forced swim), with marked variations along the rostro-caudal axis of the arcuate nucleus. In contrast, only a small population of agouti-related protein neurons in this brain region was activated. These neuronal activation patterns were correlated with behavioral reactions. Both stressors suppressed feeding and induced anxiety-like behavior in the elevated plus-maze test, as reflected by a reduction in the percentage of entries and time spent in the open arms. Central pretreatment with SHU9119, a melanocortin receptor antagonist, dose dependently attenuated the anorectic and anxiogenic effects elicited by acute restraint or forced swim. These results indicate that the melancortinergic pathway can be rapidly recruited by acute emotional stress, and that activation of melanocortin signaling is involved in mediating stress-induced anorexia and anxiety.
Figures


Similar articles
-
Melanocortin-4 receptor in the medial amygdala regulates emotional stress-induced anxiety-like behaviour, anorexia and corticosterone secretion.Int J Neuropsychopharmacol. 2013 Feb;16(1):105-20. doi: 10.1017/S146114571100174X. Epub 2011 Dec 16. Int J Neuropsychopharmacol. 2013. PMID: 22176700 Free PMC article.
-
Inhibition of immobilization stress-induced anorexia, behavioral deficits, and plasma corticosterone secretion by injected leptin in rats.Stress. 2013 May;16(3):353-62. doi: 10.3109/10253890.2012.736047. Epub 2012 Nov 5. Stress. 2013. PMID: 23035922
-
Loss of hypothalamic response to leptin during pregnancy associated with development of melanocortin resistance.J Neuroendocrinol. 2009 May;21(5):449-56. doi: 10.1111/j.1365-2826.2009.01862.x. J Neuroendocrinol. 2009. PMID: 19302191
-
The melanocortin pathway and control of appetite-progress and therapeutic implications.J Endocrinol. 2019 Apr 1;241(1):R1-R33. doi: 10.1530/JOE-18-0596. J Endocrinol. 2019. PMID: 30812013 Free PMC article. Review.
-
The leptin-dependent and -independent melanocortin signaling system: regulation of feeding and energy expenditure.J Endocrinol. 2007 Apr;193(1):1-9. doi: 10.1677/JOE-06-0144. J Endocrinol. 2007. PMID: 17400797 Review.
Cited by
-
The Interaction Between POMC rs2071345 Polymorphism and Alcohol Dependence in Anxiety Symptoms Among Chinese Male Problem Drinkers.Front Psychiatry. 2022 May 3;13:878960. doi: 10.3389/fpsyt.2022.878960. eCollection 2022. Front Psychiatry. 2022. PMID: 35592377 Free PMC article.
-
Loss of melanocortin-4 receptor function attenuates HPA responses to psychological stress.Psychoneuroendocrinology. 2014 Apr;42:98-105. doi: 10.1016/j.psyneuen.2014.01.010. Epub 2014 Jan 22. Psychoneuroendocrinology. 2014. PMID: 24636506 Free PMC article.
-
Cognitive and neural correlates of depression-like behaviour in socially defeated mice: an animal model of depression with cognitive dysfunction.Int J Neuropsychopharmacol. 2011 Apr;14(3):303-17. doi: 10.1017/S1461145710000945. Epub 2010 Aug 24. Int J Neuropsychopharmacol. 2011. PMID: 20735879 Free PMC article.
-
Hypothalamic Agouti-Related Peptide mRNA is Elevated During Natural and Stress-Induced Anorexia.J Neuroendocrinol. 2015 Sep;27(9):681-91. doi: 10.1111/jne.12295. J Neuroendocrinol. 2015. PMID: 26017156 Free PMC article.
-
Effect of barodenervation on cardiovascular responses elicited from the hypothalamic arcuate nucleus of the rat.PLoS One. 2012;7(12):e53111. doi: 10.1371/journal.pone.0053111. Epub 2012 Dec 27. PLoS One. 2012. PMID: 23300873 Free PMC article.
References
-
- Seeley RJ, Drazen DL, Clegg DJ. The critical role of the melanocortin system in the control of energy balance. Annu Rev Nutr. 2004;24:133–149. - PubMed
-
- Lu XY. Role of central melanocortin signaling in eating disorders. Psychopharmacol Bull. 2001;35:45–65. - PubMed
-
- MacNeil DJ, Howard AD, Guan X, Fong TM, Nargund RP, Bednarek MA, Goulet MT, Weinberg DH, Strack AM, Marsh DJ, Chen HY, Shen CP, Chen AS, Rosenblum CI, MacNeil T, Tota M, MacIntyre ED, VanderPloeg LH. The role of melanocortins in body weight regulation: opportunities for the treatment of obesity. Eur J Pharmacol. 2002;440:141–157. - PubMed
-
- Adan RA, Szklarczyk AW, Oosterom J, Brakkee JH, Nijenhuis WA, Schaaper WM, Meloen RH, Gispen WH. Characterization of melanocortin receptor ligands on cloned brain melanocortin receptors and on grooming behavior in the rat. Eur J Pharmacol. 1999;378:249–258. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

