A systematic review of commercial serological antibody detection tests for the diagnosis of extrapulmonary tuberculosis
- PMID: 17675320
- PMCID: PMC2094240
- DOI: 10.1136/thx.2006.075754
A systematic review of commercial serological antibody detection tests for the diagnosis of extrapulmonary tuberculosis
Abstract
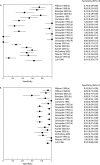
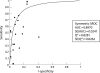
Conventional diagnostic tests for tuberculosis have several limitations and are often unhelpful in establishing the diagnosis of extrapulmonary tuberculosis. Although commercial serological antibody based tests are available, their usefulness in the diagnosis of extrapulmonary tuberculosis is unknown. A systematic review was conducted to assess the accuracy of commercial serological antibody detection tests for the diagnosis of extrapulmonary tuberculosis. In a comprehensive search, 21 studies that reported data on sensitivity and specificity for extrapulmonary tuberculosis were identified. These studies evaluated seven different commercial tests, with Anda-TB IgG accounting for 48% of the studies. The results showed that (1) all commercial tests provided highly variable estimates of sensitivity (range 0.00-1.00) and specificity (range 0.59-1.00) for all extrapulmonary sites combined; (2) the Anda-TB IgG kit showed highly variable sensitivity (range 0.26-1.00) and specificity (range 0.59-1.00) for all extrapulmonary sites combined; (3) for all tests combined, sensitivity estimates for both lymph node tuberculosis (range 0.23-1.00) and pleural tuberculosis (range 0.26-0.59) were poor and inconsistent; and (4) there were no data to determine the accuracy of the tests in children or in patients with HIV infection, the two groups for which the test would be most useful. At present, commercial antibody detection tests for extrapulmonary tuberculosis have no role in clinical care or case detection.
Conflict of interest statement
Competing interests: KW is co‐inventor on a number of patents relating to mycobacterial antigens which may be used for serological assays. All rights have been assigned to Statens Serum Institut.
References
-
- Centers for Disease Control and Prevention Reported tuberculosis in the United States, 2005. Atlanta: US Department of Health and Human Services, CDC, 200664
-
- World Health Organization TB epidemiology and surveillance workshop, 2006. http://www.who.int/tb/surveillanceworkshop/trend_analysis/increasing_dec... (accessed 6 October 2006)
-
- Iseman M D. Tuberculosis in relation to HIV and AIDS. In: Iseman MD, ed. A clinician's guide to tuberculosis. Philadelphia: Lippincott Williams and Wilkins, 2000207–209.
-
- Hopewell P C. Tuberculosis and other mycobacterial diseases. In: Mason RJ, Broaddus VC, Murray JF, Nadel JA, eds. Murray and Nadel's textbook of respiratory medicine. 4th edn. Philadelphia: Saunders, 2005984–1026.
-
- Talavera W, Lessnau K‐D K L, Handwerger S. Extrapulmonary tuberculosis. In: Friedman LN, ed. Tuberculosis. 1st edn. Boca Raton: CRC Press, 1994113–151.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical