Diatom-derived carbohydrates as factors affecting bacterial community composition in estuarine sediments
- PMID: 17675437
- PMCID: PMC2075028
- DOI: 10.1128/AEM.00551-07
Diatom-derived carbohydrates as factors affecting bacterial community composition in estuarine sediments
Abstract
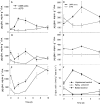
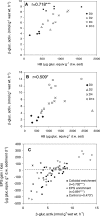
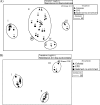
Microphytobenthic biofilms in estuaries, dominated by epipelic diatoms, are sites of high primary productivity. These diatoms exude large quantities of extracellular polymeric substances (EPS) comprising polysaccharides and glycoproteins, providing a substantial pool of organic carbon available to heterotrophs within the sediment. In this study, sediment slurry microcosms were enriched with either colloidal carbohydrates or colloidal EPS (cEPS) or left unamended. Over 10 days, the fate of these carbohydrates and changes in beta-glucosidase activity were monitored. Terminal restriction fragment length polymorphism (T-RFLP), DNA sequencing, and quantitative PCR (Q-PCR) analysis of 16S rRNA sequences were used to determine whether sediment bacterial communities exhibited compositional shifts in response to the different available carbon sources. Initial heterotrophic activity led to reductions in carbohydrate concentrations in all three microcosms from day 0 to day 2, with some increases in beta-glucosidase activity. During this period, treatment-specific shifts in bacterial community composition were not observed. However, by days 4 and 10, the bacterial community in the cEPS-enriched sediment diverged from those in colloid-enriched and unamended sediments, with Q-PCR analysis showing elevated bacterial numbers in the cEPS-enriched sediment at day 4. Community shifts were attributed to changes in cEPS concentrations and increased beta-glucosidase activity. T-RFLP and sequencing analyses suggested that this shift was not due to a total community response but rather to large increases in the relative abundance of members of the gamma-proteobacteria, particularly Acinetobacter-related bacteria. These experiments suggest that taxon- and substrate-specific responses within the bacterial community are involved in the degradation of diatom-derived extracellular carbohydrates.
Figures


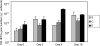
Similar articles
-
Different Types of Diatom-Derived Extracellular Polymeric Substances Drive Changes in Heterotrophic Bacterial Communities from Intertidal Sediments.Front Microbiol. 2017 Feb 27;8:245. doi: 10.3389/fmicb.2017.00245. eCollection 2017. Front Microbiol. 2017. PMID: 28289404 Free PMC article.
-
Rapid bacterial colonization of low-density polyethylene microplastics in coastal sediment microcosms.BMC Microbiol. 2014 Sep 23;14:232. doi: 10.1186/s12866-014-0232-4. BMC Microbiol. 2014. PMID: 25245856 Free PMC article.
-
Community structure of Archaea and Bacteria in a profundal lake sediment Lake Kinneret (Israel).Syst Appl Microbiol. 2007 Apr;30(3):239-54. doi: 10.1016/j.syapm.2006.05.004. Epub 2006 Jul 18. Syst Appl Microbiol. 2007. PMID: 16857336
-
Marine polysaccharide networks and diatoms at the nanometric scale.Int J Mol Sci. 2013 Oct 9;14(10):20064-78. doi: 10.3390/ijms141020064. Int J Mol Sci. 2013. PMID: 24113585 Free PMC article. Review.
-
Modelling the functioning of a coupled microphytobenthic-EPS-bacterial system in intertidal mudflats.Mar Environ Res. 2019 Sep;150:104754. doi: 10.1016/j.marenvres.2019.104754. Epub 2019 Jul 3. Mar Environ Res. 2019. PMID: 31299542 Review.
Cited by
-
A doubling of microphytobenthos biomass coincides with a tenfold increase in denitrifier and total bacterial abundances in intertidal sediments of a temperate estuary.PLoS One. 2015 May 11;10(5):e0126583. doi: 10.1371/journal.pone.0126583. eCollection 2015. PLoS One. 2015. PMID: 25961719 Free PMC article.
-
Viral Impact on Prokaryotic and Microalgal Activities in the Microphytobenthic Biofilm of an Intertidal Mudflat (French Atlantic Coast).Front Microbiol. 2015 Nov 10;6:1214. doi: 10.3389/fmicb.2015.01214. eCollection 2015. Front Microbiol. 2015. PMID: 26617575 Free PMC article.
-
Cross-domain diversity effects: linking diatom species richness, intraspecific richness, and biomass production to host-associated bacterial diversity.ISME Commun. 2024 Mar 29;4(1):ycae046. doi: 10.1093/ismeco/ycae046. eCollection 2024 Jan. ISME Commun. 2024. PMID: 39165397 Free PMC article.
-
Seasonal variation in β-glucosidase-producing culturable bacterial diversity in a monsoon-influenced tropical estuary.Environ Monit Assess. 2019 Oct 24;191(11):662. doi: 10.1007/s10661-019-7818-0. Environ Monit Assess. 2019. PMID: 31650269
-
Climate change and the microbiology of the Antarctic Peninsula region.Sci Prog. 2008;91(Pt 2):203-17. doi: 10.3184/003685008X332534. Sci Prog. 2008. PMID: 18717370 Free PMC article.
References
-
- Abdullahi, A. S., G. J. C. Underwood, and M. R. Gretz. 2006. Extracellular matrix assembly in diatoms (Bacillariophyceae). V. Environmental effects on polysaccharide synthesis in the model diatom, Phaeodactylum tricornutum. J. Phycol. 42:363-378.
-
- Admiraal, W., H. Peletier, and T. Brouwer. 1984. The seasonal succession patterns of diatom species on an intertidal mudflat: an experimental analysis. Oikos 42:30-40.
-
- Alavi, M., T. Miller, K. Erlandson, R. Schneider, and R. Belas. 2001. Bacterial community associated with Pfiesteria-like dinoflagellate cultures. Environ. Microbiol. 3:380-396. - PubMed
-
- Bartlett, M. S. 1936. The information available in small samples. Proc. Cambridge Philos. Soc. 32:560-566.
-
- Bellinger, B. J., A. S. Abdullahi, M. R. Gretz, and G. J. C. Underwood. 2005. Biofilm polymers: relationship between carbohydrate biopolymers from estuarine mudflats and unialgal cultures of benthic diatoms. Aquat. Microb. Ecol. 38:169-180.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

