Granulocyte-colony stimulating factor promotes liver repair and induces oval cell migration and proliferation in rats
- PMID: 17681181
- PMCID: PMC3130597
- DOI: 10.1053/j.gastro.2007.05.018
Granulocyte-colony stimulating factor promotes liver repair and induces oval cell migration and proliferation in rats
Abstract
Background and aims: Hepatic regeneration is a heterogeneous phenomenon involving several cell populations. Oval cells are considered liver stem cells, a portion of which derive from bone marrow (BM). Recent studies have shown that granulocyte-colony stimulating factor (G-CSF) may be effective in facilitating liver repair. However, it remains unclear if G-CSF acts by mobilizing BM cells, or if it acts locally within the liver microenvironment to facilitate the endogenous restoration program. In the present study, we assessed the involvement of G-CSF during oval cell activation.
Methods: Dipeptidyl-peptidase-IV-deficient female rats received BM transplants from wild-type male donors. Four weeks later, rats were subjected to the 2-acetylaminofluorene/partial hepatectomy model of oval cell-mediated liver regeneration, followed by administration of either nonpegylated G-CSF or pegylated G-CSF. Control animals did not receive further treatments after surgery. The magnitude of oval cell reaction, the entity of BM contribution to liver repopulation, as well as the G-CSF/G-CSF-receptor expression levels were evaluated. In addition, in vitro proliferation and migration assays were performed on freshly isolated oval cells.
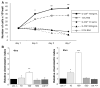
Results: Oval cells were found to express G-CSF receptor and G-CSF was produced within the regenerating liver. G-CSF administration significantly increased both the magnitude of the oval cell reaction, and the contribution of BM to liver repair. Finally, G-CSF acted as a chemoattractant and a mitogen for oval cells in vitro.
Conclusions: We have shown that G-CSF facilitates hepatic regeneration by increasing the migration of BM-derived progenitors to the liver, as well as enhancing the endogenous oval cell reaction.
Figures


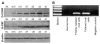

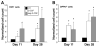




Similar articles
-
Bone marrow-derived hepatic oval cells differentiate into hepatocytes in 2-acetylaminofluorene/partial hepatectomy-induced liver regeneration.Gastroenterology. 2007 Mar;132(3):1077-87. doi: 10.1053/j.gastro.2007.01.001. Epub 2007 Jan 5. Gastroenterology. 2007. PMID: 17383429
-
Ischemia/reperfusion injury promotes and granulocyte-colony stimulating factor inhibits migration of bone marrow-derived stem cells to endometrium.Stem Cells Dev. 2012 Dec 10;21(18):3324-31. doi: 10.1089/scd.2011.0193. Epub 2012 Aug 16. Stem Cells Dev. 2012. PMID: 22897736 Free PMC article.
-
Granulocyte-colony-stimulating factor stimulation of bone marrow mesenchymal stromal cells promotes CD34+ cell migration via a matrix metalloproteinase-2-dependent mechanism.Stem Cells Dev. 2012 Nov 20;21(17):3162-72. doi: 10.1089/scd.2012.0048. Epub 2012 Jul 24. Stem Cells Dev. 2012. PMID: 22651889 Free PMC article.
-
Establishment of cancer cell lines from rat hepatocholangiocarcinoma and assessment of the role of granulocyte-colony stimulating factor and hepatocyte growth factor in their growth, motility and survival.J Hepatol. 2009 Jul;51(1):77-92. doi: 10.1016/j.jhep.2009.02.022. Epub 2009 Apr 5. J Hepatol. 2009. PMID: 19446912 Free PMC article.
-
Bone marrow progenitors are not the source of expanding oval cells in injured liver.Stem Cells. 2004;22(6):1049-61. doi: 10.1634/stemcells.22-6-1049. Stem Cells. 2004. PMID: 15536195
Cited by
-
Predictors of steroid non-response and new approaches in severe alcoholic hepatitis.Clin Mol Hepatol. 2020 Oct;26(4):639-651. doi: 10.3350/cmh.2020.0196. Epub 2020 Oct 1. Clin Mol Hepatol. 2020. PMID: 33053936 Free PMC article.
-
Hepatic Regeneration in Cirrhosis.J Clin Exp Hepatol. 2022 Mar-Apr;12(2):603-616. doi: 10.1016/j.jceh.2021.08.029. Epub 2021 Sep 4. J Clin Exp Hepatol. 2022. PMID: 35535091 Free PMC article. Review.
-
Current Perspectives Regarding Stem Cell-Based Therapy for Liver Cirrhosis.Can J Gastroenterol Hepatol. 2018 Jan 29;2018:4197857. doi: 10.1155/2018/4197857. eCollection 2018. Can J Gastroenterol Hepatol. 2018. PMID: 29670867 Free PMC article. Review.
-
Efficacy of Granulocyte Colony-Stimulating Factor in Acute on Chronic Liver Failure: A Systematic Review and Survival Meta-Analysis.J Clin Med. 2023 Oct 16;12(20):6541. doi: 10.3390/jcm12206541. J Clin Med. 2023. PMID: 37892679 Free PMC article. Review.
-
Stimulation of oval cell and hepatocyte proliferation by exogenous bombesin and neurotensin in partially hepatectomized rats.World J Gastrointest Pathophysiol. 2011 Dec 15;2(6):146-54. doi: 10.4291/wjgp.v2.i6.146. World J Gastrointest Pathophysiol. 2011. PMID: 22180848 Free PMC article.
References
-
- Shackel NA, Rockey DC. Stem cells and liver disease: promise laced with confusion and intrigue. Gastroenterology. 2004;127:346–348. - PubMed
-
- Lowes KN, Croager EJ, Olynyk JK, Abraham LJ, Yeoh GC. Oval cell-mediated liver regeneration: role of cytokines and growth factors. J Gastroenterol Hepatol. 2003;18:4–12. - PubMed
-
- Libbrecht L, Desmet V, Van Damme B, Roskams T. Deep intralobular extension of human hepatic progenitor cells correlates with parenchymal inflammation in chronic viral hepatitis: can progenitor cells migrate? J Pathol. 2000;192:373–378. - PubMed
-
- Crosbie OM, Reynolds M, McEntee G, Traynor O, Hegarty JE, O’Farrelly C. In vitro evidence for the presence of hematopoietic stem cells in the adult human liver. Hepatology. 1999;29:1193–1198. - PubMed
-
- Petersen BE, Bowen WC, Patrene KD, Mars WM, Sullivan AK, Murase N, Boggs SS, Greenberger JS, Goff JP. Bone marrow as a potential source of hepatic oval cells. Science. 1999;284:1168–1170. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

