Dynamics of Neisseria gonorrhoeae attachment: microcolony development, cortical plaque formation, and cytoprotection
- PMID: 17682045
- PMCID: PMC2044525
- DOI: 10.1128/IAI.00687-07
Dynamics of Neisseria gonorrhoeae attachment: microcolony development, cortical plaque formation, and cytoprotection
Abstract
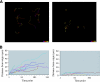
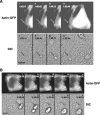
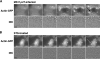
Neisseria gonorrhoeae is the bacterium that causes gonorrhea, a major sexually transmitted disease and a significant cofactor for human immunodeficiency virus transmission. The retactile N. gonorrhoeae type IV pilus (Tfp) mediates twitching motility and attachment. Using live-cell microscopy, we reveal for the first time the dynamics of twitching motility by N. gonorrhoeae in its natural environment, human epithelial cells. Bacteria aggregate into microcolonies on the cell surface and induce a massive remodeling of the microvillus architecture. Surprisingly, the microcolonies are motile, and they fuse to form progressively larger structures that undergo rapid reorganization, suggesting that bacteria communicate with each other during infection. As reported, actin plaques form beneath microcolonies. Here, we show that cortical plaques comigrate with motile microcolonies. These activities are dependent on pilT, the Tfp retraction locus. Cultures infected with a pilT mutant have significantly higher numbers of apoptotic cells than cultures infected with the wild-type strain. Inducing pilT expression with isopropyl-beta-D-thiogalactopyranoside partially rescues cells from infection-induced apoptosis, demonstrating that Tfp retraction is intrinsically cytoprotective for the host. Tfp-mediated attachment is therefore a continuum of microcolony motility and force stimulation of host cell signaling, leading to a cytoprotective effect.
Figures


References
-
- Achtman, M., M. Neibert, B. A. Crowe, W. Strittmatter, B. Kusecek, E. Weyse, M. J. Walsh, B. Slawig, G. Morelli, A. Moll, et al. 1988. Purification and characterization of eight class 5 outer membrane protein variants from a clone of Neisseria meningitidis serogroup A. J. Exp. Med. 168:507-525. - PMC - PubMed
-
- Ayala, P., J. S. Wilbur, L. M. Wetzler, J. A. Tainer, A. Snyder, and M. So. 2005. The pilus and porin of Neisseria gonorrhoeae cooperatively induce Ca2+ transients in infected epithelial cells. Cell. Microbiol. 7:1736-1748. - PubMed
-
- Belmokhtar, C. A., J. Hillion, and E. Segal-Bendirdjian. 2001. Staurosporine induces apoptosis through both caspase-dependent and caspase-independent mechanisms. Oncogene 20:3354-3362. - PubMed
-
- Bernard, B., T. Fest, J. L. Pretet, and C. Mougin. 2001. Staurosporine-induced apoptosis of HPV positive and negative human cervical cancer cells from different points in the cell cycle. Cell Death. Differ. 8:234-244. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

