Regional distribution of sodium-dependent excitatory amino acid transporters in rat spinal cord
- PMID: 17684893
- PMCID: PMC2031954
- DOI: 10.1080/10790268.2007.11753935
Regional distribution of sodium-dependent excitatory amino acid transporters in rat spinal cord
Abstract
Background/objective: The excitatory amino acid transporters (EAATs), or sodium-dependent glutamate transporters, provide the primary mechanism for glutamate removal from the synaptic cleft. EAAT distribution has been determined in the rat brain, but it is only partially characterized in the spinal cord.
Methods: The regional anatomic distribution of EAATs in spinal cord was assessed by radioligand autoradiography throughout cervical, thoracic, and lumbar cord levels in female Sprague-Dawley rats. EAAT subtype regional distribution was evaluated by inclusion of pharmacologic transport inhibitors in the autoradiography assays and by immunohistochemistry using subtype-specific polyclonal antibodies to rat GLT1 (EAAT2), GLAST (EAAT1), and EAAC1 (EAAT3) rat transporter subtypes.
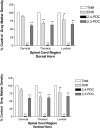
Results: [3H]-D-Aspartate binding was distributed throughout gray matter at the 3 spinal cord levels, with negligible binding in white matter. Inclusion of pharmacologic transport inhibitors indicates that the EAAT2/ GLT1 subtype represents 21% to 40% of binding. Both EAAT1/GLAST and EAAT3/EAAC1 contributed the remainder of binding. Immunoreactivity to subtype-specific antibodies varied, depending on cord level, and was present in both gray and white matter. All 3 subtypes displayed prominent immunoreactivity in the dorsal horn. EAAT3/EAAC1 and to a lesser extent EAAT1/GLAST immunoreactivity also occurred in a punctate pattern in the ventral horn.
Conclusions: The results indicate heterogeneity of EAAT distribution among spinal cord levels and regions. The presence of these transporters throughout rat spinal cord suggests the importance of their contributions to spinal cord function.
Figures

Similar articles
-
Comparison of Na+-dependent glutamate transport activity in synaptosomes, C6 glioma, and Xenopus oocytes expressing excitatory amino acid carrier 1 (EAAC1).Mol Pharmacol. 1996 Mar;49(3):465-73. Mol Pharmacol. 1996. PMID: 8643086
-
Semi-quantitative distribution of excitatory amino acid (glutamate) transporters 1-3 (EAAT1-3) and the cystine-glutamate exchanger (xCT) in the adult murine spinal cord.Neurochem Int. 2020 Nov;140:104811. doi: 10.1016/j.neuint.2020.104811. Epub 2020 Aug 5. Neurochem Int. 2020. PMID: 32768484
-
Excitatory amino acid transporter 1 and 2 immunoreactivity in the spinal cord in amyotrophic lateral sclerosis.Acta Neuropathol. 2000 Aug;100(2):138-44. doi: 10.1007/s004019900159. Acta Neuropathol. 2000. PMID: 10963360
-
GLAST But Not Least--Distribution, Function, Genetics and Epigenetics of L-Glutamate Transport in Brain--Focus on GLAST/EAAT1.Neurochem Res. 2015 Dec;40(12):2461-72. doi: 10.1007/s11064-015-1605-2. Epub 2015 May 14. Neurochem Res. 2015. PMID: 25972039 Review.
-
Excitatory Amino Acid Transporters as Therapeutic Targets in the Treatment of Neurological Disorders: Their Roles and Therapeutic Prospects.Neurochem Res. 2025 Apr 29;50(3):155. doi: 10.1007/s11064-025-04400-3. Neurochem Res. 2025. PMID: 40299102 Review.
Cited by
-
Molecular Anatomy of Synaptic and Extrasynaptic Neurotransmission Between Nociceptive Primary Afferents and Spinal Dorsal Horn Neurons.Int J Mol Sci. 2025 Mar 6;26(5):2356. doi: 10.3390/ijms26052356. Int J Mol Sci. 2025. PMID: 40076973 Free PMC article. Review.
-
Aquaporin-4-binding autoantibodies in patients with neuromyelitis optica impair glutamate transport by down-regulating EAAT2.J Exp Med. 2008 Oct 27;205(11):2473-81. doi: 10.1084/jem.20081241. Epub 2008 Oct 6. J Exp Med. 2008. PMID: 18838545 Free PMC article.
-
Targeted Overexpression of Astrocytic Endothelin-1 Attenuates Neuropathic Pain by Upregulating Spinal Excitatory Amino Acid Transporter-2.J Mol Neurosci. 2015 Sep;57(1):90-6. doi: 10.1007/s12031-015-0581-y. Epub 2015 May 21. J Mol Neurosci. 2015. PMID: 25994587
-
Role of Glutamatergic Excitotoxicity in Neuromyelitis Optica Spectrum Disorders.Front Cell Neurosci. 2019 Apr 12;13:142. doi: 10.3389/fncel.2019.00142. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31031597 Free PMC article. Review.
-
A toll-like receptor 9 antagonist restores below-level glial glutamate transporter expression in the dorsal horn following spinal cord injury.Sci Rep. 2018 Jun 7;8(1):8723. doi: 10.1038/s41598-018-26915-2. Sci Rep. 2018. PMID: 29880832 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
