Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment
- PMID: 17687264
- PMCID: PMC4753804
- DOI: 10.1038/sj.npp.1301521
Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment
Abstract
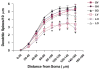
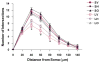
Dystrophic changes in dendrites of cortical neurons are present in several neuro-psychiatric disorders, including schizophrenia. The mechanisms that account for dendritic changes in the prefrontal cortex (PFC) in schizophrenia are unclear. Cognitive deficits in schizophrenia have been linked to compromised cortical dopamine function, and the density of the PFC dopamine innervation is decreased in schizophrenia. We determined if 6-hydroxydopamine lesions of the ventral tegmental area that disrupt the PFC dopamine innervation cause dystrophic changes in cortical neurons. Three weeks post-operatively we observed a marked decrease in basal dendritic length and spine density of layer V pyramidal cells in the prelimbic cortex; no change was seen in neurons of the motor cortex. We then examined rats in which the PFC dopamine innervation was lesioned and 3 weeks later were started on chronic treatment with an atypical (olanzapine) or typical (haloperidol) antipsychotic drug. Olanzapine but not haloperidol reversed lesion-induced changes in PFC pyramidal cell dendrites. These data suggest that dopamine regulates dendritic structure in PFC neurons. Moreover, the findings are consistent with a decrease in cortical dopaminergic tone contributing to the pathological changes in the cortex of schizophrenia, and suggest that the progressive cortical loss in schizophrenia may be slowed or reversed by treatment with atypical antipsychotic drugs.
Conflict of interest statement
The author(s) declare that, except for income received from my primary employer, no financial support or compensation has been received from any individual or corporate entity over the past 3 years for research or professional service and there are no personal financial holdings that could be perceived as constituting a potential conflict of interest, except for the following: part of this work was supported by an investigator-initiated grant from Eli Lilly & Co. AYD is a consultant to Eli Lilly. In addition, AYD derives support from the NIH and National Parkinson Foundation.
Figures


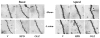
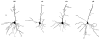
References
-
- Akil M, Pierri JN, Whitehead RE, Edgar CL, Mohila C, Sampson AR, et al. Lamina-specific alterations in the dopamine innervation of the prefrontal cortex in schizophrenic subjects. Am J Psychiatry. 1999;156:1580–1589. - PubMed
-
- Antonova E, Sharma T, Morris R, Kumari V. The relationship between brain structure and neurocognition in schizophrenia: a selective review. Schizophr Res. 2004;70:117–145. - PubMed
-
- Arnsten AF. Adrenergic targets for the treatment of cognitive deficits in schizophrenia. Psychopharmacology (Berl) 2004;174:25–31. - PubMed
-
- Arnt J, Skarsfeldt T. Do novel antipsychotics have similar pharmacological characteristics? A review of the evidence. Neuropsychopharmacology. 1998;18:63–101. - PubMed
-
- Ballesteros-Yanez I, Ambrosio E, Benavides-Piccione R, Perez J, Torres I, Miguens M, et al. The effects of morphine self-administration on cortical pyramidal cell structure in addictionprone lewis rats. Cereb Cortex. 2007;17:238–249. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

