Control of morphogenetic cell movements in the early zebrafish myotome
- PMID: 17689522
- PMCID: PMC2723113
- DOI: 10.1016/j.ydbio.2007.06.008
Control of morphogenetic cell movements in the early zebrafish myotome
Abstract
As the vertebrate myotome is generated, myogenic precursor cells undergo extensive and coordinated movements as they differentiate into properly positioned embryonic muscle fibers. In the zebrafish, the "adaxial" cells adjacent to the notochord are the first muscle precursors to be specified. After initially differentiating into slow-twitch myosin-expressing muscle fibers, these cells have been shown to undergo a remarkable radial migration through the lateral somite, to populate the superficial layer of slow-twitch muscle of the mature myotome. Here we characterize an earlier set of adaxial cell behaviors; the transition from a roughly 4x5 array of cuboidal cells to a 1x20 stack of elongated cells, prior to the migration event. We find that adaxial cells display a highly stereotypical series of behaviors as they undergo this rearrangement. Furthermore, we show that the actin regulatory molecule, Cap1, is specifically expressed in adaxial cells and is required for the progression of these behaviors. The requirement of Cap1 for a cellular apical constriction step is reminiscent of similar requirements of Cap during apical constriction in Drosophila development, suggesting a conservation of gene function for a cell biological event critical to many developmental processes.
Figures


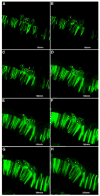
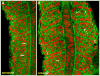


References
-
- Afonin B, Ho M, Gustin JK, Meloty-Kapella C, Domingo CR. Cell behaviors associated with somite segmentation and rotation in Xenopus laevis. Dev Dyn. 2006;235:3268–79. - PubMed
-
- Amacher SL, Kimmel CB. Promoting notochord fate and repressing muscle development in zebrafish axial mesoderm. Development. 1998;125:1397–406. - PubMed
-
- Barresi MJ, Stickney HL, Devoto SH. The zebrafish slow-muscle-omitted gene product is required for Hedgehog signal transduction and the development of slow muscle identity. Development. 2000;127:2189–99. - PubMed
-
- Baum B, Perrimon N. Spatial control of the actin cytoskeleton in Drosophila epithelial cells. Nat Cell Biol. 2001;3:883–90. - PubMed
-
- Benlali A, Draskovic I, Hazelett DJ, Treisman JE. act up controls actin polymerization to alter cell shape and restrict Hedgehog signaling in the Drosophila eye disc. Cell. 2000;101:271–81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

